Cloning and Expression Characters of Chalcone Isomerase Gene from Camellia nitidissima
ZHOU Xing-wen1,2 , LI Ji-yuan1*, FAN Zheng-qi1, YIN Heng-fu1
1Research Institute of Subtropical Forestry, Chinese Academy of Forestry, Fuyang, 311400, China
2Yu Lin Normal University, Guangxi Zhuang Autonomous Region, Yulin, 537000, China
* Author for correspondence, Email: jiyuan_li@126.com; Tel/Fax: 86 571 63346372
Introduction
Plants of Section Chrysantha Chang belongs to Genus Camellia L., Theaceae, evergreen shrubs or small trees. The model species of this section is C. nitidissima Chi(Zhang et al, 1998). This species is well known for its golden flowers and is reputed to be the ‘Queen of Theaceae’, ‘Panda of the vegetable kingdom’ in China and ‘yellow camellia of the imagination abroad’(Liang,1992). As an important parent to breed new yellow camellias, C. nitidissima Chi has been paid much attention by camellia breeders since it was reported (Chen et al,1987;Parks 1989;2000;Chen et al,1994;Nishimoto,2004).
Previous researches showed that the main composition of floral pigmentation from C. nitidissima Chi is quercetin-7-glucoside(Miyajima et al,1985), which belongs to the flavonoids. In the pathway of flavonoids biosynthesis, CHI is regarded as an important enzyme which can catalyse the chalone to naringenin by isomerization, and the naringenin can be catalysed by the subsequence enzymes to form the others substance(Tanaka et al,2008). The CHI genes have been cloned in some species such as Zea mays (Grotewold et al,1994) L., Petunia hybrid (van Tunen et al,1988)Vilm., Medicago sativa(McKhann et al,1994) L. etc., and the researches about changing the flower’s color by regulating the CHI with genetic engineering were reported (LING et al,2009).
In this report, we cloned the CnCHI and analysed its expression characters for the purpose of finding the role of CHI in the biosynthesis of flavonoid pigment in C. nitidissima Chi and providing correlative theory for breeding the newly yellow camellia by genetic engineering.
Material and Methods
Species sampling
C. nitidissima Chi was planted in the experimental greenhouse of Research Institute of Subtropical Forestry, Chinese Academy of Forestry. Flowers in different developmental stages (Table 1) were collected and stored in a silver paper bag separately to extract the total RNA for RT-PCR and relative quantitative real time PCR, frozen in liquid nitrogen immediately and stored at -80°C before extracting.
Cloning full length cDNA of CnCHI by RT-PCR and RACE
Total RNA was extracted from flower petals, using the plant pillar RNAout kit (TIANDZ, Beijing, China) and each RNA samples was treated by DNase, using pillar DNA-free (TIANDZ, Beijing, China). Total RNA was reverse transcribed into cDNA using avian myeloblastosis virus (AMV) reverse transcriptase (Fermentas, Ontario, Canada). The primers SP/AP (Table 1) were designed by Primer 5.0 according to the conserved domain of CHI from other species in Genbank to amplify the homologous sequence, and sequenced by Beijing Genomics Institute (BGI). After blasting the amplified sequence in Genbank and analyzing its homology to CHI genes from others species, the sequences GSP1/GSP2 were used as primers to obtain the 3’ and 5’ ends of CHI from C. nitidissima Chi by the methods of 3’ and 5’ RACE. Link the homologous, 3’ and 5’ sequences together to form a complete full length sequence by the software Vector NTI 8.0. Then, amplify the full length cDNA by the primers P3/P4 designed according to the 3’-UTR and 5’-UTR of the spelled sequence, sequenced by BGI, blasted in Genbank to ensure it belongs to the gene CHI.
Bioinformatics analysis of CnCHI and its coding protein
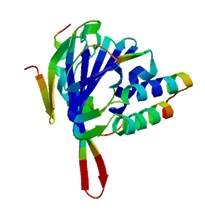
Homology of CnCHI was analysed by BLAST from NCBI (http://www.ncbi.nlm.nih.gov) and the ORF was found using the tool ORF Finder from NCBI. The amino acid composition, molecular weight, theoretical isoelectric point and instability index etc. were analysed by the ProtParam tool(http://web.expasy.org/). The alpha-helix, beta turn, random coil and extend strand were analysed by ProtScale, HNN tool from expasy(Http://cn.expasy.org). The three-dimensional structure of the predicted protein was constructed by the tool SWISS-MODEL using the Homology modelling method. The spelling of full length cDNA and the creation of homology tree were completed by the Vector NTI Suit 8.0 and DNAMAN software.
Measuring spatio-temporal expression of CnCHI by relative quantitative real time PCR
Total RNA was reverse transcribed into cDNA using avian myeloblastosis virus (AMV) reverse transcriptase (Fermentas, Ontario, Canada) and the included random primer and oligo-dT primers. Gene-specific primers were designed on basis of the homology and were listed in following Table 2. Ribosomal 18S RNA (GenBank accession number: U42815.1) was used as an endogenous control. Two step methods was applied with SYBR green fluorescent dye (TARAKA, Dalian, China) and the program for amplification included 30 s polymerase activation at 95°C followed by 40 cycles of 95°C for 5 s and 60°C for 31 s. The instrument is ABI PRISM 7300 real time PCR system (ABI, Carlsbad, United States). Every sample was repeated three times alone, the data were analyzed by the method of 2-ΔΔCT (Livak et al,2001).
Results
Cloning full length cDNA of CnCHI
Table 1 Description of floral buds at different developmental stages of C. nitidissima
| Stages | Morphology of Flower buds | ||
| Buds diameter (cm) | Status | Color | |
| 1 | 0.8±0.1 | Closed | Light green |
| 2 | 1.1±0.1 | Closed | Yellow green |
| 3 | 1.4±0.1 | Closed | Light yellow |
| 4 | 2.2±0.2 | half-opening | Golden |
| 5 | 4.0±0.5 | Fully opening | Golden |
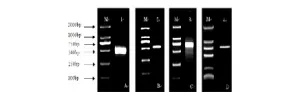
A cDNA fragment of 517 bp was amplified with primers SP/AP by PCR using the petal cDNA of C. nitidissima Chi as template (Fig.1-A-1). With the help of RACE and primers GSP1/GSP2, 3’ and 5’ ends were amplified (Fig.1-B-2, 1-C-3). The full length cDNA of CnCHI was isolated by the primers P3/P4 (Fig.1-D-4).
Nucleotide sequence analysis of CnCHI
ORF Finder of NCBI revealed that CnCHI contains an ORF of 693 bp, a 5’-UTR of 56 bp, a 3’-UTR of 204 bp. Furthermore, a Polyadenylation signal sequence AATAA which is a typical signal in plants and Poly (A) were included in CnCHI (Fig. 2). These results indicated that CnCHI is a complete gene sequence.
 |
 |
|
Fig. 1 The 1% agarose gel electrophoresis of CnCHI fragments |
Fig. 2 Full length of CnCHI and its predicted protein. |
 |
Note: 1 Red panes show initiation codon and termination codon; 2 Black line shows the tail signal;3 Dotted line shows poly-A. |
| The three dimensional structure of CnCHI |
Table 2 Primers’ sequence and their purpose
| Primer name | Primer sequence and use | |
| Sequence ( 5’ to 3’) | purpose | |
| SP | CACGGCTATCGGAGTGTACCTC | Amplifying the conserved sequence |
| AP | GGCTTCCGGTTTCTCCTTCA | |
| GSP1 | GAAAGACGGTTCCTTACCTGAGACTGGG | Amplifying the 3’ end |
|
GSP2 |
CCAGTCTCAGGTAAGGAACCGTCTTTCG | Amplifying the 5’ end |
| P3 | AAGAGGTGCTGAGAGAGAAAGAG | Amplifying the full length of CnCHI |
|
P4 |
CACGTAAACCAACTATGCCAAT | |
| Y1 | CGTCCAAGCCCTTCTTCCTC | Real time PCR |
| Y2 | TGCAGTCTTGCCCTTCCACTT | |
| 18SF | GACTCAACACGGGGAAACTTACC | Real time PCR(control) |
| 18SR | CAGACAAATCGCTCCACCAAC | |
Protein structure and functional sites analysis of CnCHI
ORF Finder analysis revealed the ORF of CnCHI located in the 57~749bp region, which encoding a protein of 230 amino acids. ProtParam analysis showed that the protein molecular weight is 24.677kD, theoretical isoelectric point of 5.41, total number atoms of 3504, molecular formula C1120H1763N277O340S4, without signal peptide. Instability coefficient is 47.82, which indicates that CnCHI is an unstable hydrophilic protein. The secondary structure of CnCHI is mainly composed of random coil, accounting for 43.04%; followed by α-helix, 38.7%; extend chain accounted for 18.26%, which is consistent with the predicted three dimensional structure(Fig. 3). Function sites analysis showed that CnCHI contains two conserved hydrogen bonding sites Thr (T52) and Tyr (Y110) (Fig. 4-A), two active catalytic sites Asn (N117) and Ser (S194) (Jez et al, 2002) (Fig. 4-B), seven substrate binding sites Arg40, Gly41, Leu42, Phe51, Ile54, Lys113, Val114[16], these sites are highly conserved and the relative positions are not changed. The alignment of CnCHI amino acids with other species showed that CnCHI has 99% homology with C. sinesis L. and has the homology more than 75% with the species from Rosaceae, Ericaceae and Solanaceae.
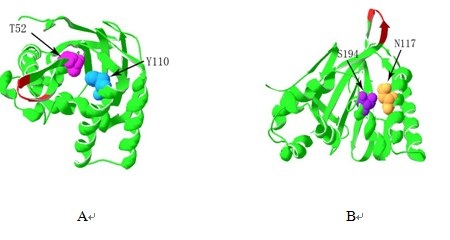
Fig. 4 The function sites of CnCHI
Expression characters of CnCHI
Expression of CnCHI at different flower developmental stages: The expression of CnCHI at different developmental stages was detected by relative quantitative real time PCR method, shown in Fig.8. The result indicated that the highest expression level of CnCHI was at stage 2, almost three times higher than the expression of stage 1, the lowest level was at stage 5. The result showed a tendency to a sharp increase at the early stage and then decreased slowly (Fig. 5).
Expression of CnCHI in different floral organs: The result of relative quantitative real time PCR to study the CnCHI expression level in different floral organs was shown in Fig.5. It can be seen that the highest expression level of CnCHI was in the pistil, which followed by stamens, the transcript of CnCHI in sepals and petals were almost at the same level (Fig. 6).
|
|
|
|
Fig. 5 The relative expression of CnCHI at different stages |
Fig. 6 The relative expression of of CnCHI in different parts of flowers |
Discussion
In this research, the CnCHI was obtained from the petals of C. nitidissima Chi by the method of RT-PCR and RACE. The bioinformatics analysis of CnCHI indicates that this gene contains an ORF of 693bp encoding 230 amino acids and the secondary structure of CnCHI is mainly composed of random coil. The higher homology of amino acids with C. sinensis L. than other species suggests that CnCHI is very conservative among the same genus, which means that CnCHI may be used to obtain the yellow flower by suppressing the others camellias’ CHI expression. The results that two conserve hydrogen bonding sites Thr (T52) and Tyr (Y110), two active catalytic sites Asn (N117) and Ser (S194) (Jez et al, 2000; 2002) and seven substrate binding sites located in CnCHI shows that CnCHI may be active and involved in the pigment biosynthesis of C. nitidissima Chi, which means CnCHI may play an important role in regulating the biosynthesis of pigment.
The analysis of relative quantitative real time PCR showed that the expression level of CnCHI during the developmental stages of flower buds exhibits the tendency to a sharp increase at the early stage and then decreases slowly later. This phenomenon may indicate that CnCHI expresses mainly at the early developmental stage of flower buds of C. nitidissima Chi, which is agreed with that CHI is an important enzyme which is involved in flavonoids biosynthesis of plants (Tanaka et al, 2008). The tendency is that the highest expression level of CnCHI was in the pistil, which is followed by stamens indicating that the expression of CnCHI has definite expression specificity, which is accordant with the researches of McKhann(Tunen et al, 1988) and ZHOU Jun’s(Zhou et al, 2009). These results show that CnCHI is mainly expressed in the parts that are young and grow vigorously. It can be concluded that the expression of CnCHI has an important effect on the other flavonoids according to the results.
In conclusion, CnCHI is an important gene which may play an important role on the pigment biosynthesis of C. nitiddisima Chi. Research showed that suppressing the activity of CHI can make the flowers yellow (Takamura et al, 1995; Itoh et al, 2002; Nishihara, 2005). So, it is worth researching the CnCHI more deeply to enable the breeding the new yellow camellias by genetic engineering.
Acknowledgment
This work was financially supported by following research projects (2011DFA30490; No. 200704028 and No. 2007-4-04).
References
ZHANG H., REN S., 1998. Flora of China, vol. 49, 3rd fascicule, Beijing, Science Press.
LIANG S. 1992.Golden camellia, Beijing, China Forestry Publishing House.
CHEN J., 1987.Fourteen years of golden camellia breeding. Journal of BeiJing Forestry University, No. 9: 315–320.
Parks C.R., 1989.The hybrids of Camellia chrysantha, International Camellia Journal, No.21: 76-79.
Parks C., 2000. Breeding progress with yellow camellias. American Camellia Yearbook, 2000: 9-10.
CHEN J., CHEN J., ZHAO S., 1994. Interspecific cross breeding for new yellow camellia., Journal of BeiJing Forestry University, No.16: 315–320.
Nishimoto S., Hashimoto F., Shimizu K., Sakata Y.. 2004. Petal colcoration of interspecific hybrids between Camellia chrysantha and japonica, No.2004:189-191.
Miyajima, I. Uemoto S., Sakata Y. et al, 1985. Yellow pigment of Camellia chrysantha flowers, American Camellia Yearbook, 1985,71-80.
Tanaka Y., Sasaki N., Ohmiya A., 2008. Biosynthesis of plant pigments: anthocyanins, betalains and carotenoids. The Plant Journal, No.54:733-749.
Grotewold E., Peterson T., 1994. Isolation and characterization of a maize gene encoding chalcone flavonone isomerase. Mol Gen Genet,No. 242: 1-8.
van Tunen A. J., Koes R. E., Spelt C. E., van der Krol A. R., Stuitje R.S., Mol J. N.M., 1988. Cloning of the two chalcone flavanone isomerase genes from Petunia hybrida: coordinate,light-regulated and differential expression of flavonoid genes. EMBO J, No. 7:1257-1263.
McKhann H. I., Hirsch A. M., 1994. Isolation of chalcone synthase and chalcone isomerase cDNAs from alfalfa (Medicago sativa L.): highest transcript levels occur in young roots and root tips. Plant Mol Biol, No. 24:767-777.
LING W., GUO H., 2009. Anthocyanin of plants. Beijing, Science Press.
Livak K. J., Schmittgen T. D., 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2 (-Delta Delta C (T)) method, Methods, No. 25: 402-408.
Jez J. M., Bowman M. E.,2002. Role of hydrogen bonds in the reaction mechanism of chalcone isomerase, Biochemistry No. 41: 5168-5176.
Jez J. M., Bowman M. E., Dixon R. A., Noel J. P., 2000. Structure and mechanism of the evolutionarily unique plant enzyme chalcone isomerase. Nature America Inc, No. 9 :786-791.
Tanaka Y., Sasaki N., Ohmiya A., 2008. Biosynthesis of plant pigments: anthocyanins, betalains and carotenoids. The Plant Journal, vol.54, pp.733-749.
ZHOU J., YAO Q., PENG R., Xiong A., CAI B., Xu J., et al, 2009. Cloning and expression of CHI of Kyoho Grape by semi-quantity RT-PCR, Acta Botanica Boreali-Occidentalia Sinica, No. 29:1723-1729.
Takamura T., Tomihama T., Miyajima I., 1995. Inheritance of yellow-flowered characteristic and yellow pigments in diploid cyclamen (Cyclamen persicum Mill.) cultivars. Scientia Hort, No. 64:55-63.
Itoh Y., Higeta D., Suzuki A., Yoshida H., Ozeki, Y., 2002. Excision of transposable elements from the chalcone isomerase and dihydroflavonol 4-reductase genes may contribute to the variegation of the yellow-flowered carnation (Dianthus caryophyllus). Plant Cell Physiol, No.43: 578-585.
Nishihara M., Nakatsuka T., Yamamura S., 2005. Flavonoid components and flower color change in transgenic tobacco plants by suppression of chalcone isomerase gene. FEBS Lett, No. 579:6074-6078.
Web design by Tribal Systems


