Sanitary status of camellias growing at the urban parks and gardens from the Pazos in Galicia (NW Spain)
Mansilla, J.P., Pérez-Otero, R., Aguín, O., Pintos, C., Rial, C., Chaves, M. and Salinero, C.
Estación Fitopatolóxica do Areeiro, Deputación de Pontevedra, Subida a la Robleda s/n, 36153 Pontevedra, Spain. E-mail: pedro.mansilla@depo.es
Introduction
Camellia species, and their hybrids and cultivars, are widely disseminated in the Galicia region (NW Spain). Ornamental camellias, mainly Camellia japonica and to a lesser extent C. reticulata and C. sasanqua, were introduced into Galicia in the 18th century. They are currently being propagated in several nurseries specializing in ornamentals and can also be found in public parks and historical gardens (pazos) in Galicia. However, at present there are several diseases and different species of arthropod pests that are damaging these plants.
Several fungi have been reported to cause camellia root rot including Armillaria mellea, Phytophthora species (Mansilla et al., 1991, 1998, 2001a, b) and Rosellinia necatrix (Aguín et al., 2002). Camellia species are also affected by a number of aerial fungal diseases namely Botrytis cinerea, Ciborinia camelliae, Glomerella cingulata, Meliola camelliae, Pestalotia guepinii, Phomopsis camelliae-japonicae and Phytophthora ramorum (Mansilla et al., 2001a, b). Of these, Ciborinia camelliae (Mansilla et al., 1999) and Phytophthora ramorum (Pintos et al., 2003) are included in the European Plant Protection Organization (EPPO) Pest lists (EPPO A2 list) and they were detected, for the first time in Spain, in Galicia. There are also several references to trunk diseases found on Camellia spp. of which Camellia dieback and canker caused by Glomerella cingulata is the most cited wood disease. Some species of Botryosphaeriaceae have already been reported for Camellia, but it was not until 2012 when Neofusicoccum species (Neofussicocum luteum and N. parvum, Botryosphaeriaceae family) were identified on camellias in Spain (Pintos et al., 2012). Finally, several species of insects and mites are listed in the bibliography by country (Johnson & Lyon, 1991; Ferrari et al., 1999). Most are sucking insects (scales, aphids), Coleoptera, Lepidoptera and Eriophyioidea mites. Up to now, representatives of all these groups, except Lepidoptera, have been found in Galicia (Mansilla et al., 2003a).
Material and methods
In a continuing survey during 2013, six historic gardens from Galician Pazos and five urban public parks were sampled. A total of 170 samples (60 soils and 110 aerial Camellia parts: branches, shoots and leaves) were collected. The selected pazos were: Pazo Gandarón (Pontevedra), Pazo Lourizán (Pontevedra), Pazo Oca (A Estrada, Pontevedra), Pazo Quiñones León (Vigo, Pontevedra), Pazo Rubianes (Villagarcia, Pontevedra), Soutomaior Castle, and five urban gardens in the cities of Vigo and Pontevedra.
Identification of fungal aerial diseases
Plants showing canker, dieback and leaf spots were kept in a controlled chamber (20-25°C, 16-8h day-night, 75-80 % humidity). Fungi observed were isolated, transferred onto Potato Dextrose Agar (PDA) and incubated at 25°C in darkness. Pure cultures of the fungi isolates were obtained and identified by cultural, physiological and morphological characteristics and confirmed by molecular analysis. Fungal DNA was directly extracted from mycelia growing on plates, using the commercial kit EZNA™ Fungal DNA Miniprep Kit (Omega Bio-tek) following the short protocol. PCR reactions were carried out with primers ITS1 and ITS4 (White et al., 1990). Amplified products were sequenced and the sequences obtained were analyzed with the BLAST alignment program of the NCBI and compared with those deposited in GenBank database.
Isolation and Characterization of Phytophthora species from Rhizosphere soil
A survey of the occurrence of Phytophthora species from sixty camellia rhizosphere soils samples was conducted in the six selected Pazos, but not in the urban parks.
Rhizosphere soils were tested by flooding with distilled water and baited with young leaflets of avocado (Persea americana) and petals of Dianthus sp. (Jung et al., 1996, 2000). After incubation for 48-96 h, leaflets were examined using a light microscope for determinate the presence of Phytophthora sporangia. Leaflets with sporangia were blotted on filter paper, to remove excess water, and placed onto selective V8 agar. Petri® dishes were incubated at 22 °C in the dark. Colonies growing from the baits were examined. Phytophthora isolates were identified using morphological and physiological characters and DNA sequences.
The morphological identification was based on the colony morphology, cardinal growth temperatures, and production, morphology and dimensions of sporangia, antheridia and oogonia. The presence of hyphal swellings or chlamydospores was also examined (Erwin and Ribeiro, 1996). Colony patterns and growth characteristics were described from 10 days old culture grown at 23 °C in the dark onto V8. Formation of sporangia was induced by flooding agar discs, cut from the growing edge of 7 day-old culture grown on V8 agar, with non-sterile soil extract water and incubated at room temperature for 48-72 h in the dark (Jung et al., 1996). After incubation the dimensions, shape, papilla, and caducity of sporangia were recorded for each isolate. Gametangia were measured from colonies on V8, kept for ten or more days, as described by Brasier et al., (2004). Dimensions and characteristic features of oogonia, oospores and antheridia were recorded.
In order to confirm the identification derived from the morphological studies a subset of isolates of each species was selected for DNA sequence analysis. DNA fungal extraction was performed with the commercial kit EZNA™ Fungal DNA Miniprep Kit (Omega Bio-tek) following the short protocol. DNA amplification and sequencing was carried out according to protocols proposed by Kroon et al. (2004) and Cooke et al. (2000), analyzing a DNA fragment corresponding to the elongation factor and a fragment of the ribosomal DNA ITS region, respectively. The sequences obtained were compared with sequences available in GenBank database using BLAST program.
Isolation of Armillaria spp. from Rhizosphere soil
A survey on the occurrence of Armillaria species from rhizosphere soils where camellia plants were growing was conducted in the pazos. Sixty soil samples were collected in the six selected pazos.
DNA extraction was performed by using the PowerSoil® DNA Isolation Kit (MoBio Laboratories, Inc.). Each fungal sample was needed to obtain 2-10 ng of DNA.
DNA amplification was performed by Nested-PCR with Ready-to-Go PCR Beads (Amersham Biosciences). Two PCR reactions were realized .The first PCR reaction was carried out with primers (ITS1/ITS4) and the second with (AR1/AR2) primers. Amplification was performed under the conditions proposed by Lochman et al. (2004). PCR products were digested with two restriction enzymes, namely Mbo I and Hinf I. The digested DNA fragments were separated by electrophoresis. The identification of Armillaria species was based on the size of digested fragments as compared to those reported by Lochman et al. (2004).
Identification of arthropod pests
Samples were examined in laboratory using a binocular magnifier to find arthropods. When mites, aphids or thrips were found, they were separated from vegetable support using an entomological needle and each specimen was placed in a receptacle with lactic acid (70%) for its clarification on a hotplate. Then, they were mounted on microscope slides with Hoyer. The procedure for samples with scales was different: first, the females were placed in glass capsules with 10% NaOH; subsequently they were washed using a mixture of acetic acid (25%), water (50%) and 70% alcohol (25%) and they were transferred to a receptacle with 70% alcohol for at least 15 minutes. Then they were dyed using chlorazol black for a few seconds and put again in 70% alcohol and later in acid fuchsin (for at least during 24 hours). The next step was alcohol dehydration: first using 70% alcohol (15 minutes), then 96% (15 minutes) and 100% alcohol (15 minutes). Finally specimens were put in Euparal essence for 15 minutes and then they were mounted on microscope slides using Euparal.
Results and discussion
Identification of fungal aerial disease
In this survey, some of the main aerial diseases that affect camellias, and that have already been recorded in Galicia (Mansilla et al., 1991, 1998, 2001a, b), were also found. Included among those fungi that affect the aerial part of the plant are Botrytis cinerea, Colletotrichum gloeosporoides, Meliola camelliae, Pestalotia guepinii and Phomopsis camelliae (Figure 1). In this survey we have also identified other aerial fungi that have more recently appeared on camellia plants in Galicia that could be Neofusicoccum parvum and Neofusicoccum luteum (Pintos et al., 2012) or Botryosphaeria dothidea and Diplodia mutila.
Botryosphaeriaceae fungi caused leaf spots (Figure 2), cankered branches and dieback. Neofusicoccum and Botryosphaeria are genera that comprise many species; they have a cosmopolitan distribution and could cause dieback and canker diseases in woody hosts (Denman et al., 2000). Neofusicoccum parvum, Neofusicoccum luteum, Botryosphaeria dothidea and Diplodia mutila are polyphagous fungi detected in multiple hosts.
 |
 |
Figure 1. Symptoms of Phomopsis on shoot Figure 2. Foliar damage caused by Botryosphariaceae
Phytophthora species isolated from Rhizosphere soil
In this survey three Phytophthora species, P. cinnamomi, P. citricola and P. cryptogea (Figure 3), were identified using morphological and physiological characters. Identity was confirmed by DNA sequences analysis. Comparison of the sequences obtained showed 100% homology with sequences available in GenBank database for these three Phytophthora species.
P. cinnamomi form abundantly terminal or intercalary chlamydospores in the mycelium. Characteristic hyphal swellings are also produced (Figure 3A). P. cinnamomi is a heterothallic species. Sex bodies are formed from intra-specific crossing in colonies paired on V8 using an A1 and A2 reference isolates of P. cinnamomi. Antheridia are amphigynous (Figure 3C). The sporangia in water are noncaducous and nonpapillate with shapes varying from spherical to ellipsoid (Figure 3B). Optimum temperature for growth in V8 is 24-27°C.
P. citricola is a homothallic species. Oospores are mostly plerotic and antheridia are paragynous (Figure 3F). Shapes of sporangia are highly variable and ranged from ovoid to ovo-pyriform (Figure 3E). Sporangia with double papillae and distorted shapes are common. Optimum temperature is 26-28° C.
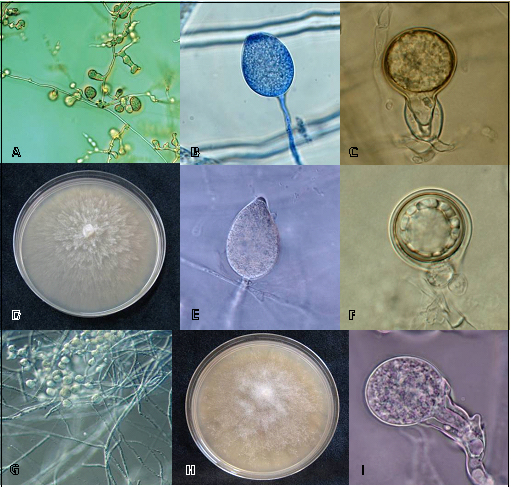 |
Figure 3. Morphological characteristics of Phytophthora species isolated from soils of Galician Pazos. A, B, C, hyphal swellings, sporangia and sex organs of P. cinnamomi. D, E, F, colony, sporangia and sex bodies of P. citricola. G, H, I, mycelium, colony and sex organ of P. cryptogea.
P. cryptogea is a heterothallic species. Sex bodies (Fig. 3I) are formed from inter-specific crossing using A1 and A2 reference isolates of P. cinnamomi. Hyphal swellings formed in chains or clusters are abundant in aqueous cultures (Fig. 3G). Sporangia are nonpapillate and persistent. Both external and internal proliferations occur. Optimum temperature for growth in V8 is 25°C. The morphological characteristics observed in our isolates were very similar to the Phytophthora species descriptions reported in literature (e.g. Erwin and Ribeiro, 1996; Brasier et al., 2004; Gallegly and Hong, 2008).
In this survey, P. cinnamomi was isolated in three rhizosphere soils (5%) belonging to two pazos (Pazo Lourizán and Pazo Rubianes). P. citricola was isolated in two rhizosphere soils (3.3%) belonging to Pazo Lourizán and Pazo Oca and P. cryptogea was isolated in one soil (1.7%) belonging to Pazo Quiñones de León (Table 1).
Only P. cinnamomi had previously been detected in Camellia roots and soils in Galicia (Mansilla et al., 2001a, b). To date, P. cryptogea and P. citricola have only been reported in other ornamental hosts in Spain as Viburnum tinus, Rhododendron spp. or Buxus sempervirens (Moralejo et al., 2009). To our knowledge, this is the first report of P. cryptogea and P. citricola from rhizosphere camellia soils in Galicia.
Armillaria species isolated from Rhizosphere soil
The identification of Armillaria species was based on the size of digested fragments as compared to those reported by Lochman et al. (2004). The restriction enzyme MboI discriminate A. mellea generating a pattern of bands 235, 215, 149 and 125 bp as shown in the Figure 4A, but the restriction endonuclease MboI was not able to discriminate between the species A. cepistipes and A. gallica and in these cases the enzyme HinfI is used for differentiate this species obtaining fragments of 294, 227 69/63 and 43 bp for A. gallica (figure 4B).
Armillaria mellea (Figure 5) was isolated in a total of five rhizosphere soils belonging to three pazos and Armillaria gallica (Figure 6) was isolated in a total of eight rhizosphere soils belonging to three pazos, from a total of sixty soils analyzed (Table 1). Armillaria species are fungi that destroy the root system, resulting in progressive decay, leaf fall and reduction of growth (Mansilla et al., 1991, 1998, 2001a, b).
|
|
Armillaria gallica |
Armillaria mellea |
Phytophthora cinnamomi |
Phytophthora citricola |
Phytophthora cryptogea |
Phytophthora sp. |
|
Pazo Lourizán |
4 |
x |
1 |
1 |
x |
x |
|
Pazo Gandarón |
x |
1 |
x |
x |
x |
2 |
|
Soutomaior Castle |
x |
1 |
x |
x |
x |
x |
|
Pazo de Oca |
1 |
3 |
x |
1 |
x |
2 |
|
Pazo Quiñones de León |
3 |
x |
x |
x |
1 |
2 |
|
Pazo de Rubianes |
x |
x |
2 |
x |
x |
x |
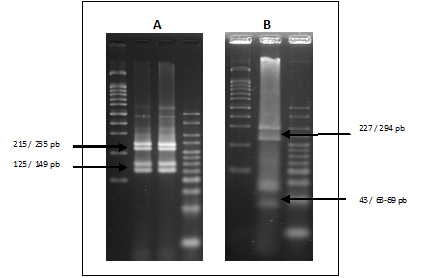 Figure 4: A, Fragments of A. mellea with enzyme MboI.
Figure 4: A, Fragments of A. mellea with enzyme MboI.
B, Fragments of A. gallica with enzyme HinfI.
 |
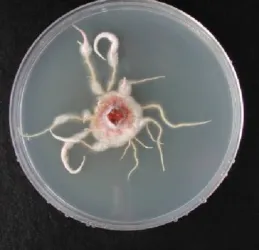 |
Figure 5. Fructification bodies of A. mellea Figure 6. Mycelia of Armillaria gallica
Arthropod pests detected in samples
Populations of insects, mites or their symptoms were detected in all samples. However, none of them was causing damage significant enough to compromise the viability of the plants. They could only affect its vigour and its aesthetic value.
There was a greater diversity of insects than mite species. However, most of the specimens of the class Insecta belong to some order of sucking insects. The main group of insects identified were the Homoptera, namely the superorder Coccoidea (5 species) and Aphidoidea (1 species), although populations of Thysanoptera and damages caused by Coleoptera Curculionidae have also been detected. Regarding the mites, the number of Eriophyoidea was higher than any other superfamily.
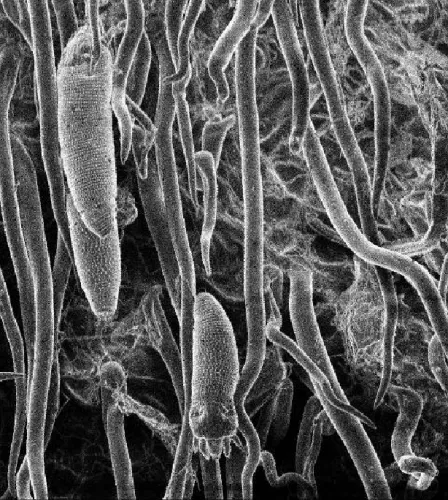
Only one pest was detected in all the survey points: Cosetacus camelliae Keifer (Prostigmata: Eriophyidae: Cecidophyinae). The camellia bud mite lives under foliar and floral scales. This species affects the edges of scales from the beginning of its development, causing its necrosis. With high populations, floral buds dry out and remain initially on the plant and then they fall to the ground: at low infestation densities, flowers fall at the moment of its outbreak. In the survey, it was detected in 57% of all samples. The number of mites varied among the different samples: the highest value was 500 mites /sepal, and the minimum, 10 mites /sepal. With these data, plants with the highest population rates showed a high proportion of dried floral buds, while the lowest population rates showed marginal necrosis over the scales.
 |
 |
Figure 7. Cosetacus camelliae in sepal (SEM, 300 x) and symptoms of its attack
The other two species of mites detected in the survey were the tetranychid Panonychus ulmi Koch (14% of all samples) and the Eriophyidae Acaphylla steinwedeni Keifer. The last one was detected in only 5% of all samples, where no symptoms were observed. However, the leaves of plants affected with P. ulmi showed a peculiar tanned coloration. The eriophyioid mite Calacarus carinatus Green and the tarsonemid mite Polyphagotarsonemus latus Banks, previously detected in Galicia (Mansilla et al., 2003b; Pérez-Otero et al., 2007), were not found in samples collected in this survey.
The second species in occurrence was the aphid Toxoptera aurantii Boyer de Fonscolombe. It was detected in 45% of all samples. It is the most frequent aphid found on camellias in Galicia, and the only one detected in these surveys, where its colonies were not very abundant. For this reason only 15 samples showed symptoms: curled and wrinkled leaves and sooty mould over the honeydew Toxoptera secretes. None of the plants showed signs of lack of vigour.
The occurrence of these three Coccoidea species was also important: Chloropulvinaria floccifera Westwood and Coccus hesperidium Linnaeus from the family Coccidae, and Hemiberlesia rapax Comstock from the family Diaspididae. Chloropulvinaria floccifera was detected in 21% of all samples. Usually, only females with their ovisacs or rests were observed and in only 14 samples, rests of fumagine associated with honeydew were found. Coccus hesperidium was found in 16% of all samples. Generally very few individuals, about 2-4, formed the colonies. Therefore, they did not cause symptoms. In addition, in nearly half of the positive samples, some individuals were found with an emergence hole of some parasitoid. Hemiberlesia rapax was the next scale species in order of abundance: it was detected in 27% of collected samples. This result was somewhat surprising, since in surveys carried out in the area a few years ago, it was not very abundant, which probably indicates that it is increasing its population. It was found on the leaves, petioles and in the scales of floral buds, although previously it was only located on leaves. However, no symptoms associated with their presence were found, as the colonies were always small in number.
The other species of scale insects found in surveys had a more limited presence: Planococcus citri Risso (Coccoidea: Pseudococcidae) was found in only 5% of all samples and Parlatoria camelliae Comstock (Coccoidea: Diaspididae) was found in only one sample. Samples associated with P. citri did not show any symptoms: nevertheless, the sample with P. camelliae showed leaves with important chlorotic lesions around scales of the females.
Also, an occasional presence of the thrips Heliothrips haemorrhoidalis Bouché was found in 6% of all samples and always on leaves, not on flowers. The symptom of its outbreak is silver or leaden coloured discoloration on the underside of leaves, where populations are normally found. Nevertheless, this symptom was not observed in all samples collected.
Finally, hardly any damage was detected in samples due to attack by Curculionidae, whose presence in Camellia is normal.
Conclusion
The results of this study indicate that although the number of genera and species of fungi and arthropods identified is high, these have only been isolated in a few specimens of Camellia sampled. So we can conclude that the health status of the camellias in Galicia is good. In the coming years, new surveys will be required for checking if the phytosanitary status of Galician camellias continued stable.
Acknowledgements
We thank Ana Garcia Servia, Raquel Romero, Bernabé Peón and Ana Belén Castro for their skillful technical assistance.
References
Aguín, O., Sainz, M.J., Salinero, C., Mansilla J.P. 2002. First report of Rosellinia necatrix on Camellia in Spain. Plant Disease 86: 813.
Brasier, C.M., Kirk, S.A., Delcan, J., Cooke, D.E.L., Jung, T. & Man in´t Veld, A. (2004). Phytophthora alni sp. nov. and its variants: designation of emerging heteroploid hybrid pathogens spreading on Alnus trees. Mycological Research, 108 (10): 1172-1184
Cooke D.E.L, Drenth A., Duncan J.M., Wagels G. & Brasier C.M. (2000). A molecular phylogeny of Phytophthora and related Oomycetes. Fungal Genetics and Biology, 30, 17-32
Denman, S., Crous, P.W., Taylor, E., Kang, J., Pascoe, I. and Wingfield, J. 2000. An overview of the taxonomic history of Botryospaheria, and re-evaluation of its anamorphs based on morphology and ITS rDNA phylogeny. Studies in Mycology 45: 129-140
Erwin, D.C., Ribeiro, O.K. (Eds.) (1996). Phytophthora Diseases Worldwide. APS Press, St. Paul.
Ferrari, M., Menta, A., Marcon, E., Montermini A. 1999. Malattie e parassiti delle piante da fiore, ornamentali e forestali. Edagricole. 1806 pp.
Gallegly, M. E., Hong, C.H. (Eds.) (2008). Phytophthora: Identifying Species by Morphology and DNA Fingerprints. APS Press, St. Paul.
Johnson, W.T., Lyon, H.H., 1991. Insects that Feed on Trees and Shrubs. Second Edition Revised. Comstock Publishing Associates. 560 pp.
Jung, T., Blaschke, H. & Neumann, P. (1996). Isolation, identification and pathogenicity of Phytophthora species from declining oak stands. European Journal of forest Pathology, 26: 253-272
Jung, T., Blaschke, H.,& Obwald, W., 2000. Involvement of Phytophthora species in Central European oak decline and the effect of site factors on the disease. Plant Pathology, 49: 706-718
Kroon, L.P.N.M.; Bakker, F.T.; Van Den Bosch, G.B.M.; Bonants, P.J.M. & Flier W.G. (2004). Phylogenetic analysis of Phytophthora species based on mitochondrial and nuclear DNA sequences. Fungal genetics and Biology, 41: 766- 782
Lochman J, Sery O, Mikes V. 2004. The rapid identification of European Armillaria species from soil samples by nested PCR. FEMS Microbiol Lett. 237: 105-110.
Mansilla Vázquez, P., Pérez Otero, R., Salinero Corral, C., López Villarreal, M.J., Barrote Meira, M.C. 2003a. Presencia y distribución de ácaros eriófidos en camelias del sur de Galicia y norte de Portugal. Bol. San. Veg. Plagas, 29: 35-41.
Mansilla Vázquez, P., Salinero Corral, C., Pérez Otero, R., Pintos Varela, C. 2003b. Plagas, enfermedades y fisiopatías de la camelia. 2ª edición. Xunta de Galicia. 107 pp.
Mansilla J.P., Aguín O., Vela P., Salinero C. 2001a. Fungal diseases affecting Camellia in Spain. En: American Camellia Yearbook 2000. Ed. American Camellia Society, Fort Valley (Georgia, USA), pp. 18-21.
Mansilla Vázquez, J. P.; Pintos Varela, C. y Salinero Corral, M. C. 2001b. Enfermedades y fisiopatías frecuentes sobre Camellia en Galicia: daños y control. En: La Camellia. Servicio de Publicaciones. Excma. Diputación Provincial de Pontevedra. España. 115-125.
Mansilla Vázquez, J. P.; Salinero Corral, M. C. y Pintos Varela, C. 1999. Ciborinia camelliae Kohn. Fungo causante do murchamento da flor da camelia. Tríptico editado por la Xunta de Galicia.
Mansilla Vázquez, J. P.; Salinero Corral, M. C. y Vela Fernández, M. P. 1998. Enfermedades y fisiopatías frecuentes sobre Camelia en Galicia: daños y control. Phytoma España 104: 38-42.
Mansilla Vázquez J. P.; Salinero Corral M. C.; Fernández Lorenzo J. L. 1991. Phytosanitary problems detected on Camellia in Galicia (Spain). Official Publication of the American Camellia Society. The Camellia Journal 47 (1): 4-6.
Moralejo, E., Pérez¬-Sierra, A.M., Álvarez, L.A., Belbahri, L., Lefort, F., Descals, E., 2009. Multiple alien Phytophthora taxa discovered on diseased ornamental plants in Spain. Plant Pathology 58 (1): 100-110.
Pérez-Otero, R., Mansilla-Vázquez, J.P., Salinero-Corral, M.C. 2007. First Report of the Broad Mite Polyphagotarsonemus latus Banks on Camellia japonica in Spain. American Camellia Yearbook: 52-56.
Pintos, C.; Redondo, V.; Chaves, M..; Rial, C., Aguín, O.; Mansilla, J.P. and Salinero, C. 2012. Camellia japonica dieback canker by Neofusicoccum luteum and N. parvum in Spain. International Camellia Journal. 44: 126-128
Pintos Varela, C., Mansilla Vázquez, J. P. y Aguín Casal, O. 2003. First Report of Phytophthora ramorum on Camellia japonica in Spain. Plant Disease Volume 87, Nº11, Pag: 1396
White, T.J, Bruns T, Lee, S. and Taylor, J .1990. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. En: Innis M.A., Gelfand D.H., Sninsky J.J. y White T.J. (eds) PCR protocols: a guide to methods and application, pp 315-322. San Diego,CA,USA: Academic Press.
Web design by Tribal Systems
