The Study on DNA of ChuXiong population of Camellia reticulata Lindl. of China by FTIR
Lu Qiu1, 2*,[1]RenmingLiu3, Ying Chen4, Chenbo Cai1, Peng Liu5, Haiyan Yang1, Suguo Fang1, Minzhen Si 3
1. Department of Chemistry and Life Science, Chuxiong Normal University,
Chuxiong 675000, P. R. China;
2. Institute of Bio-resources Research and Utilization in Central Yunnan,
Chuxiong 675000, P. R. China;
3. Department of Physics and Electronics Science, Chuxiong Normal University,
Chuxiong 675000, P. R. China;
4. Department of Geophysical Sciences of Chuxiong Normal University,
Chuxiong 675000, P. R. China;
5. Department of Mathematics of Chuxiong Normal University,
Chuxiong 675000, P. R. China
[1]*Corresponding author.Tel: 0086-8783100507;fax: 0086-8783139505.
E-mail address: qiulu@cxtc.edu.cn
1. Introduction
Fourier Transform Infrared (FTIR) spectroscopy has the advantage of being a highly sensitive, fast, non-destructive method of producing spectra using only small amounts of sample material[1, 2]. For these reasons, it has been widely applied to qualitative and quantitative analyses in chemistry, biology, medicinal chemistry, disease diagnosis and environmental science etc [3-7]. In recent years, with the rapid development of chemometrics, computer science and molecular biology, the studies on DNA molecules by using FTIR is particularly eye-catching [8, 9]. FTIR can bring structural information of the entire DNA molecule in a single spectrum as a “snapshot” of all conformations, including possible conformational substrates present in the sample [1,10]. Thus it can monitor subtle variations of DNA structures and conformation. So, IR spectroscopy was extensively applied to study the variations in the formation of the farinose ring, the carbohydrate -phosphate DNA backbone, and the G–C, A–T base pairing of DNA double helix [11-13]. This formation is from entire genome, DNA fragment, nucleotide, the interaction of medicament with DNA etc. [14-18].
Camellias belong to the plants of Camellia genus of Theaceae. They have a very high ornamental and economic value. They are famous throughout the world and a Chinese traditional flower. China has the widest distribution of wild camellias in the world. Yunnan province is the distribution center of C. reticulata Lindl. in China. ChuXiong is one of the important distribution centers of C. reticulata Lindl. in the Yunnan province. ChuXiong population of C. reticulata Lindl. is unique. It has been identified and described by camellia experts, and much of this information is available on the internet under Chinese flowers and plants. It is famous for having the biggest flowers, the brightest colors and the most ancient tree.
At present, PCR-based molecular markers are often used to identify Camellia varieties, such as RAPD, AFLP,ISSR and ITS etc. [19-22]. Although PCR-based molecular markers methods have been found to be accurate and reliable, this technology requires complicated procedures, specialized techniques and laboratory equipment. Time-consuming and costly, these methods are not sufficiently convenient and rapid for application anywhere and by anyone. So, spectroscopy-based identifying methods for plants, animal and microbe by FTIR of DNA has been reported recently [5, 8, 12, 15, 17, 18].
There are a few reports about FTIR of Camellia. There are not any reports about FTIR of DNA of Camellia or C. reticulata Lindl. Shen JB (2008) investigated FTIR spectra of sixty-five leaf samples from Camellia sect., Oleifera sect., Paracamellia sect, and sect. Camellia (Theaceae). The FTIR method was used directly on the leaf samples, which were freeze-dried, pulverized and rubbed. The results showed that the FTIR spectra of Camellia has fingerprint-like patterns that are highly typical for different taxa, and the hierarchical dendrogram based on principal component analysis (PCA) of FTIR data was constructed by the Ward’s method using a Euclidean distance measure. Sixty-five leaf samples were divided into two main clusters. The hierarchical dendrogram shows that the relationship between species is in agreement with most of the previous proposed morphological classifications. It was thought that this technique could be widely used for identification and classification of other taxa when a standard spectrum is available [23], but the FTIR spectra were from the mixture of leaf, not at a biomacromolecule, or molecular level and the accuracy is not consistent.
In this study, the author extracted DNA from the leaf, and obtained FTIR spectra of DNA of ChuXiong populationof C. reticulata Lindl. A new method of identifying and classifying different varieties of C. reticulata Lindl. at DNA molecular level was set up. This method possesses high resolution, accuracy and repeatability, and is quick and easy.
2. Materials and methods
2.1. Materials
Material was collected from ZiXi mountain natural preservation areas at ChuXiong city in YunNan province of China, and included ten kinds of the ChuXiong populationof C. reticulataLindl, shown in Table 1. Among them, the camellia trees ‘XiangGuo’, ‘YanZhi’, ‘ChuXiong’, ‘GuoMei’, ‘ZeHe’, ‘LuChengChun’, ‘ZhiNan’ and ‘DongLin’ are over 100 years old. ‘XiangGuo’ is the oldest tree; it is over 800 years old. ‘ZiDingHongLian’ and ‘JiaoYuan’ are the youngest trees; they are only twenty years old. ‘XiangGuo’, ‘DongLin’, ‘ZiDingHongLian’, ‘JiaoYuan’ and ‘ZhiNan’ are the primary variety variants; ‘ChuXiong’, ‘GuoMei’, ‘YanZhi’, ‘ZeHe’ and ‘LuChengChun’ were formed by grafting.
2.2. DNA extracting method
DNA ofC. reticulata Lindl. was extracted by using the improved SDS-CTAB method. 0.25 g fresh leaves were taken and ground finely, then DNA extraction buffer 750 μl ( Tris-HCl 200 m mol / L、EDTA 100 m mol / L、NaCl 12.6 m mol / L ), 10 ℅ CTAB 300 μl, 10 % SDS 300 μl, and sulphur ethanol 100 μl was added. The mixture was fully blended and processed at 65Cby using water boiled for 30 minutes. It was precipitated by centrifuge for 5-10 minutes at 12000 rpm. The supernatant liquid was taken out. Equal volumetric phenol saturated by water was added and oscillated for 5 minutes. The supernatant liquid was taken out again, and equal volumetric chloroform - isoamyl alcohol (volume 24:1) was added. The extraction was repeated three times to clear away protein. Isopropyl alcohol was mixed with supernatant liquid and stored at 4°C for one night to precipitate DNA. RNA enzyme was mixed in to clear away RNA. 75 % ethanol was used to wash precipitation twice, and the DNA precipitation was taken [24].The quality and purity of the precipitation were measured by determination of the absorption ratio of 260/ 280 nm using a DMS 200 spectrophotometer (Varian Techtron Pty. Ltd.). The 260/280 ratio of DNA sample was found to be 1.81. It shows the precipitation is DNA.
DNA with the quality of 20-50 ng /ul was chosen and lyophilized using a rotary SpeedVac (ALPHA1-2LD, Christ Co., Germany) for 3 h. DNA from Camellia was scanned by FTIR spectrometer.
2.3. FTIR microspectroscopy
FTIR spectra of DNA were obtained by using FT-IR spectrometer (Spectrum 100, PerkinElmer Co., Britain) in transmission mode in the region of 4000~400cm1 with a spectral resolution of 4cm-1. The scan number was set at 16 times. 150 mg KBr and 0.5 mg DNA were mixed and ground up, then pressured into slice. In detection process, atmospheric absorptions correction and the method of KBr compression were employed. All the spectra were analyzed with the software of Spectrum V 8.0.
2.4. Data preprocessing and analysis
In this paper, Principal Component Extraction, Hierarchical Clustering and Related Analysis were employed. The data was pretreated by using smoothing, the first derivative and unitary method. Covariance matrix was used toextract the principal component, andregression was used to calculate factor score. Nine principal components were extracted and used to cluster and identified by using Cluster Analysis. The clustering method is Ward. The clustering distance was measured by using squared Euclidean distance. Origin 8.0 and PASW 18.0 software were used to make the graph and deal with data.
3. Results
3.1. DNA FTIR of ChuXiong population of C. reticulata Lindl.
DNA FTIR from ChuXiong population of C. reticulata Lindl show that there are three outstanding absorbance bands in all varieties. The first band is at 1639 cm-1. The second is at 1050 cm-1. The third is at 977cm-1. There are some bands at 1599 cm-1,1522 cm-1, 1411 cm-1, 1352 cm-1, 1293 cm-1, 1024 cm-1, 919 cm-1, 825 cm-1, shown in Fig. 1. and Table 1. It is typical of the characteristic of DNA FTIR. In the region of 1000-800 cm-1, IR bands are sensitive to the changes in the end cyclic torsion angles of the furanose ring, it reflects sugar conformation. The bands in the 1250-1000 cm-1 region are assigned to vibrations along the sugar-phosphate backbone of B-type DNA conformation.
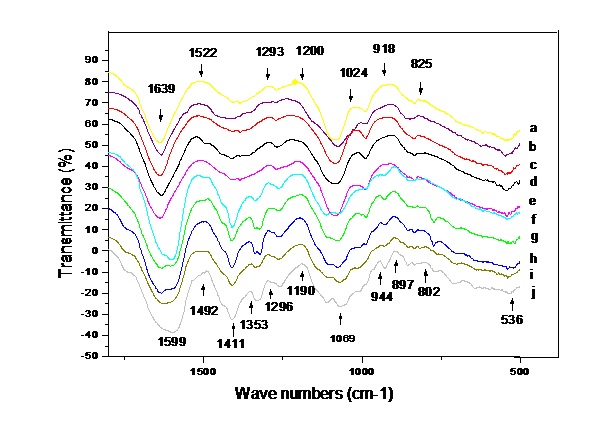
Fig.1.The absorbance spectra of DNA FTIR of ten kinds of ChuXiong population of C. reticulata Lindl. in the region of 1800~500 cm -1. The letters of the right side represent different varieties individually. Letter a. stand for ‘XiangGuo’, b. ‘ZiDingHongLian’, c. ‘YanZhi’, d ‘ChuXiong’. e. ‘GuoMei’, f. ‘JiaoYuan’, g. ‘ZeHe’, Letter h. ‘LuChengChun,’ i. ‘ZhiNan,’ j. ‘DongLin’.
The first derivative further shows the accurate wavenumbers and the obvious distinction between ten kinds of ChuXiong population of C. reticulata Lindl. ( Fig.2 ). The first derivative not only effectively cleaned noise, but also effectively enlarged valuable information, therefore the first derivative analysis is essential for DNA FTIR. The wavenumber, shape, and relative strength are different in the first derivative. It has four main characteristics. The first is that band’s shape is spikier. So, the wavenumbers is more accurate. The wavenumber of absorbance spectra is at 1665 cm-1, 1616 cm-1, 1319 cm-11340 cm-1, 1050 cm-1, 977 cm-1, 909 cm-1, 831 cm-1, 782 cm-1, 774 cm-1 in the first derivative. The second is that distinction between varieties is more obvious than the original absorbance spectra. Distinction Varieties 1, 2, 3, 4 and 5 are more similar, and varieties 6, 7, 8, 9, 10 are similar. The leading distinction is in 1616 cm-1 ~1050 cm-1. The bands at 1616 cm-1, 1050 cm-1 and 977 cm-1 are strong, and occur in all ChuXiong population of C. reticulata Lindl. The fourth is that the wavenumber changes more between different varieties. The wavenumber at 1616 cm -1 shifts from 1617 cm -1 to 1562 cm -1, shift is 15 cm -1. The wavenumber at 1050 cm -1 shifts from 1060 cm -1 to 1044 cm -1, shift is 16 cm -1. The wavenumber at 977 cm -1 shifts from 980 cm -1 to 976 cm -1, shift is 2 cm -1. It is the most steady and strongest in all bands.
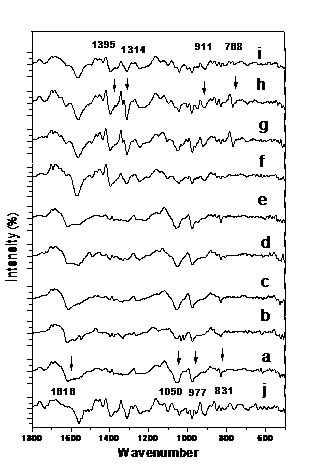
Fig.2. The first derivative of DNA FTIR of ten kinds of ChuXiong population of C. reticulata Lindl. in the region of 1800~500 cm -1.
In the region of 1000-800 cm-1, IR bands are sensitive to the changes in the end cyclic torsion angles of the furanose ring; it reflects sugar conformation [25-28]. The bands in the 1250-1000 cm-1 region are assigned to vibrations along the sugar-phosphate backbone of B-type DNA conformation [27, 31-33].
3.2. Pretreatment on the spectra data from DNA FTIR
3.2.1. Smoothing
At first, spectra were pre-treated by smoothing to 13 point: smoothing can get rid of noise. Figure 3. shows that the noise was serious in a single variety of ChuXiong population of C. reticulata Lindl, and the line is not smooth. But smoothed lines show noise has been eliminated.
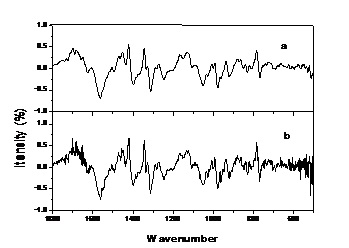
Fig.3. The first derivative of a single variety of ChuXiong population of C. reticulata Lindl.
Letter “a” stand for spectrum smoothed to 13 point. Letter “b” stand for unsmoothed spectrum.
There is serious noise in the unsmoothed first derivative of DNA FTIR. It is very serious between wavenumber of 800 cm -1~500 cm -1 ( Fig. 4. ). The noise was effectively eliminated in the smoothed first derivative ( Fig. 5. ).
 |
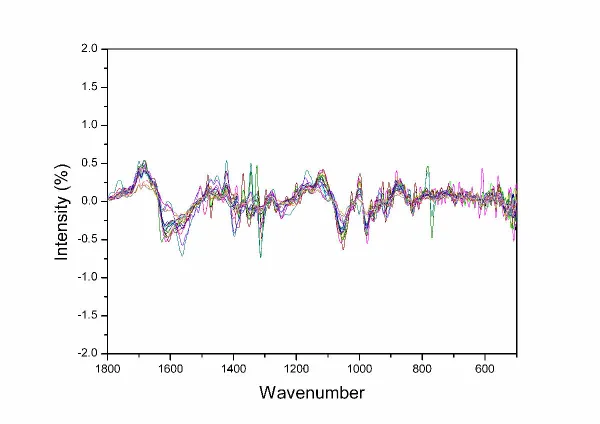 |
| Fig. 4. The first derivative not smoothed and layered of DNA FTIR of ten kinds of ChuXiong population of C. reticulata Lindl. in the region of 1800~500 cm -1. |
Fig. 5. The first derivative smoothed for 13 point and not layered of DNA FTIR of ten kinds of ChuXiong population of C. reticulata Lindl. in the region of 1800~500 cm -1. |
3.2.2. Derivativing
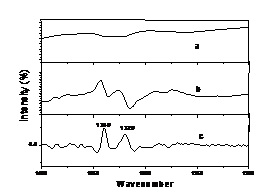
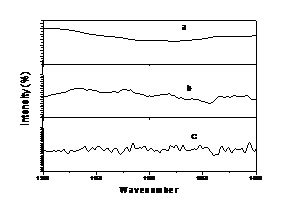
Derivativing can effectively amplify important information, so it can effectively clean noise. Figure 6. shows that the first derivative gives out the information not displayed in original absorbance spectra at 1340 cm -1 and 1320 cm -1. The second derivative further enhances information at 1340 cm -1 and 1320 cm -1 on the basis of the first derivative. Therefore, the second derivative can effectively amplify information compared with the first derivative. But Figure 7. shows that the second derivative enhances noise when it amplifies important information. So, the first derivative was used to deal with the spectra data in this investigation.
 |
 |
| Fig.6. The contrast diagram of original absorbance spectra, the first derivative and the second derivative between 1400 cm -1~1200 cm -1. |
Fig.7. The contrast diagram of original absorbance spectra, the first derivative and the second derivative between 1400 cm -1~1200 cm -1. |
[a. for original absorbance spectra, b. for the first derivative, c. for the second derivative.]
The unitary method was used by division with the band strength at 977 cm -1 to remove the influence of the DNA concentration. The band at 977 cm -1 is the most steady and strongest of all bands. It appears in all varieties, and the shift is only 2 cm -1.
3.2.3. Principal Component Extraction
Principal Component Analysis can not only effectively reduce order variables and simplify calculation, it can also effectively reduce noise and remove interference.
In this paper, Correlation matrix was used toextract the principal component, andregression was used to calculate factor score coefficient matrix. Results show that there is an obvious turning point at the ninth component. So nine principal components were extracted and used to cluster and identified by using Cluster Analysis. The cumulative percentage of nine principal components is 100 %.
3.3. Clustering analysis based on the DNA FTIR of ChuXiong population of C. reticulata Lindl.
Four DNA samples were prepared and scanned for each variety ofChuXiong population of C. reticulata Lindl. The spectra of three samples was used for clusteranalysis, and were given the number a, b and c. One sample was used for Identification analysis, and was given the number d.
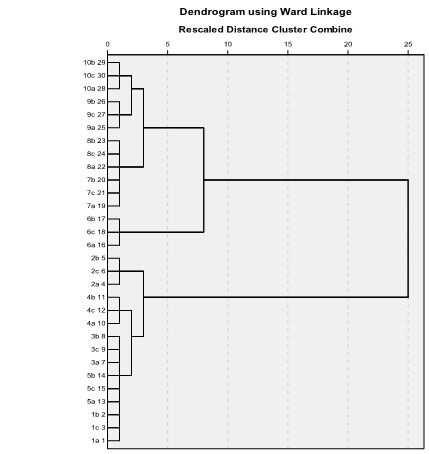
Fig. 8. Dendrogram of Hierarchical Cluster of 30 samples from 10 varieties.
The Hierarchical Cluster was used based on nine extracted principal components. The cluster method is Ward. The distance was measured by using squared Euclidean distance.
Results show that all samples can be correctly clustered. Three samples of each variety were clustered into one category. The rate of correct clustering of thirty samples is 100% (Fig. 8. and Table 2.). The three samples from variety 1 were clustered into one with the three samples from variety 5. But six samples were not hybrids. The three samples from variety 1 concentrated together. The three samples from variety 5 gathered together. It accounts for the close relationship between varieties 1and 5. The relationship between varieties 5 and 3, and between varieties 7 and 8 is closer. So, Hierarchical Cluster is very effective in the analysis of DNA FTIR.
Table 1. The result of Hierarchical Cluster of 30 samples from 10 varieties.
|
Variety Number |
Variety Name |
Sample Name |
Cluster result |
Right or False |
Right Rate |
|
1
|
‘XiangGuo’ |
1a |
1 |
Right |
100% |
|
1b |
1 |
Right |
|||
|
1c |
1 |
Right |
|||
|
2
|
‘ZiDingHong Lian’ |
2a |
2 |
Right |
100% |
|
2b |
2 |
Right |
|||
|
2c |
2 |
Right |
|||
|
3
|
‘YanZhi’ |
3a |
3 |
Right |
100% |
|
3b |
3 |
Right |
|||
|
3c |
3 |
Right |
|||
|
4
|
‘ChuXiong’ |
4a |
4 |
Right |
100% |
|
4b |
4 |
Right |
|||
|
4c |
4 |
Right |
|||
|
5
|
‘GuoMei’ |
5a |
5 |
Right |
100% |
|
5b |
5 |
Right |
|||
|
5c |
5 |
Right |
|||
|
6
|
‘JiaoYuan’ |
6a |
6 |
Right |
100% |
|
6b |
6 |
Right |
|||
|
6c |
6 |
Right |
|||
|
7 |
ZeHe |
7a |
7 |
Right |
100% |
|
|
|
7b |
7 |
Right |
|
|
7c |
7 |
Right |
|||
|
8
|
‘LuChengChun’ |
8a |
8 |
Right |
100% |
|
8b |
8 |
Right |
|||
|
8c |
8 |
Right |
|||
|
9
|
‘ZhiNan’ |
9a |
9 |
Right |
100% |
|
9b |
9 |
Right |
|||
|
9c |
9 |
Right |
|||
|
10
|
‘DongLin’ |
10a |
10 |
Right |
100% |
|
10b |
10 |
Right |
|||
|
10c |
10 |
Right |
3.4. Identifing analysis based on the DNA FTIR of ChuXiong population of C. reticulata Lindl.
The standard clustering model was established by using average spectra from each variety of ChuXiong population of C. reticulata Lindl. The first derivative was pretreated by smoothing for 13 point,
Principal Component Extraction and Hierarchical Cluster were used to set up the model. Figure 9. is the standard clustering model of ten kinds of ChuXiong population of C. reticulata Lindl.
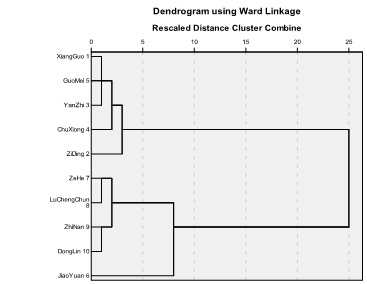
Fig. 9. The standard clustering model of ten kinds of ChuXiong population of C. reticulata Lin
|
Stage |
Cluster Combined |
Coefficients |
Stage Cluster First Appears |
Next Stage |
|||
|
Cluster 1 |
Cluster 2 |
Cluster 1 |
Cluster 2 |
||||
|
|
1 |
1 |
5 |
1.886 |
0 |
0 |
2 |
|
2 |
1 |
3 |
11.523 |
1 |
0 |
5 |
|
|
3 |
7 |
8 |
25.426 |
0 |
0 |
6 |
|
|
4 |
9 |
10 |
41.392 |
0 |
0 |
6 |
|
|
5 |
1 |
4 |
62.562 |
2 |
0 |
7 |
|
|
6 |
7 |
9 |
94.632 |
3 |
4 |
8 |
|
|
7 |
1 |
2 |
131.746 |
5 |
0 |
9 |
|
|
8 |
6 |
7 |
246.048 |
0 |
6 |
9 |
|
|
9 |
1 |
6 |
629.304 |
7 |
8 |
0 |
|
Table 2 Cluster distance coefficients of standard clustering model.
The cluster distance coefficients from Table 2 shows that the relationship is the closest between variety 1 (‘XiangGuo’) and variety 5 (‘GuoMei’), and the cluster distance coefficients is 1.886. The relationship is closer between variety 1 (‘XiangGuo’) and variety 3 (‘Yangzi’ ), and the cluster distance coefficients is 11.523. The cluster distance coefficients is 13.894 between variety 5 (‘GuoMei’) and variety 3 (‘YanZhi’). The relationship is close between variety 7 (‘ZeHe’) and variety 8 (‘LuChengChun’), and the cluster distance coefficients is 25.426. The cluster distance coefficients is 41.392 between variety 9 (‘ZhiNan’) and variety 10 (‘DongLin’).
The samples from each variety were put into standard clustering model for identification analysis. Result showed that ten samples were identified correctly. The correct clustering rate is 100% ( Fig.10. ).
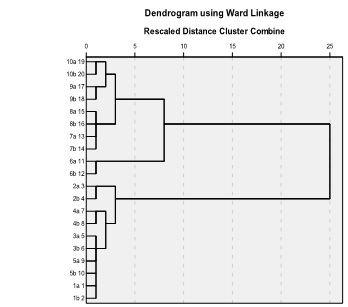
Fig.10. Hierarchical Cluster Dendrogram for identifying variety.
4. Discussion
4.1. DNA FTIR of ChuXiong population of C. reticulata Lindl.
In the region of 1000-800 cm-1, IR bands are sensitive to the changes in the end cyclic torsion angles of the furanose ring, it reflects sugar conformation. The band in the region of 840–825cm-1 is associated to the vibration modes of S-type sugar marker band in B-DNA vibration modes, which is a vibration mode localized to the C3’–C4’–C5’–O5’–P part of the sugar–phosphate backbone [25-28].
The bands in the 1250-1000 cm-1 region are assigned to vibrations along the sugar-phosphate backbone of B-type DNA conformation. The 1250cm-1 band is associated to the asymmetric stretch vibration of PO2− group of the phosphodiester-deoxyribose backbone, and the band at 1000 cm-1 is due to the symmetric stretching vibration of PO2− group [29, 30]. The bands in this region are characteristic markers for backbone conformational changes of DNA [27, 31-33].
The bands at 1297 and 1287 cm-1 aredue to the vibrations of C4-NH2 group of cytosine.1 The bands in the 1440-1330 cm-1 region reflects glycosyl bond rotation conformation. The band at 1375 cm -1 is assigned to guanosine for the glycosyl torsion angle x in anti conformation, and the band at 1354 cm -1 is due to cytidine [25,34-36].
The bands in the 1750–1500 cm-1 region are related to the G–C and A–T base pairs. 13, 31, 37 The band in the 1652-1651 cm-1 is due to C2=O2 vibrations of base-paired of cytosine bases, 1647 cm -1 band is overlapping stretching vibrations of C4=O4 of thymine, the band in the 1638-1645 cm -1 is stretching vibrations of NH2 of guanine , and the 1624 cm -1 band is associated to C4–C5=C6 ring stretch vibration of cytosine. 1503 cm -1 band is assigned to vibrations of cytosine ring [10, 25, 37].
Therefore, DNA FTIR is like a fingerprint. The fingerprint can be used as the fast identification method of the ChuXiong population of C. reticulata Lindl.
4.2. Relationship and variety origin based on the DNA FTIR of ChuXiong population of C. reticulata Lindl.
The relationship is the closest between variety 1 (‘XiangGuo’) and variety 5 (‘GuoMei’), and the cluster distance coefficients is 1.886. The cluster distance coefficients is 13.894 between variety 5 (‘GuoMei’) and variety 3 (‘YanZhi’). It was calculated based on relationship analysis that ‘GuoMei’ (variety 5) was formed by grafting the scion from the original old tree ‘XiangGuo’ ( variety 1) to a seedling. ‘YanZhi’ ( variety 3 ) was formed by grafting the scion from the trees ‘GuoMei’ ( variety 5 ) to seedling.
The relationship is close between variety 7 (‘ZeHe’) and variety 8 (‘LuChengChun’), and the cluster distance coefficients is 25.426. But they were formed by grafting. It was calculated that they were from some kind of ChuXiong population of C. reticulata Lindl. This needs further research.
The cluster distance coefficients is 41.392 between variety 9 (‘ZhiNan’) and variety 10 (‘DongLin’). But they are the primary variant varieties without grafting. They were from some kind of ChuXiong population of C. reticulata Lindl. This need further research, too.
DNA IR characteristic spectra of ten kinds of ChuXiong population of C. reticulata Lindl. were obtained. It’s of the typical characteristic of DNA FTIR. The region of 1000-800 cm-1 reflects sugar conformation. The 1250-1000 cm-1 region are assigned to vibrations along the sugar-phosphate backbone of B-type DNA conformation. DNA IR spectra are like fingerprint, and can be used to identify ten kinds of ChuXiong population of C. reticulata Lindl. The spectra data was pretreated by smoothing, the first derivative and unitary method. Nine principal components were extracted. Hierarchical Cluster was used to cluster and identify ChuXiong population of C. reticulata Lindl. The clustering method is Ward. The clustering distance was measured by using squared Euclidean distance. The standard clustering model was established by using average spectra. Result show that the correct rate of clustering and identifying is 100%. The cluster distance coefficients shows that the relationship is the closest between variety 1 (‘XiangGuo’) and variety 5 (‘GuoMei’), and the cluster distance coefficients is 1.886. The relationship is closer between variety 1 (‘XiangGuo’) and variety 3 (‘YanZhi’), and the cluster distance coefficients is 11.523. The cluster distance coefficients is 13.894 between variety 5 (‘GuoMei ‘ and variety 3 (‘YanZhi’). The relationship is close between variety 7 (‘ZeHe’) and variety 8 (‘LuChengChun’), and the cluster distance coefficients is 25.426. The cluster distance coefficients is 41.392 between variety 9 (‘ZhiNan’) and variety 10 (‘DongLin’).
Acknowledgement
The authors thank the senior engineers Fanyi Zhang, Taiqin Li, Qinwen zhao and Jinlong Yang from ChuXiong Camellia Association for providing and identifyingChuXiong population C. reticulata Lindl.
References
[1] R. Talia Kahn, K. Kimberly Fong, Brian Jordan, C. Janista Lek, Rachel Levitan, S. Patrick Mitchell, Corrina Wood, E. Mary Hatcher.BioChem. 48 (2009) 1315–1321.
[2] I. Galina Dovbeshko, Nina Ya Gridina, B. Elena Kruglova, P. Olena Pashchuk. Atlanta 53 (2000) 233–246.
[3] K. Deepak Jangir, Gunjan Tyagi, Ranjana Mehrotra, Suman Kundu. J. Mol. Struct. 969 (2010)126–129.
[4] R.M. Liua, W.Y. Tang, Y.P. Kang, M.Z. Si. Spectrochim. Acta, Part A 73 (2009) 601–607.
[5] C. Murali Krishna, G.D. Sockalingum, A. Rani Bhat, L. Venteo,Pralhad Kushtagi, M. Pluot, M. Manfait. Anal. Bioanal. Chem. 387 (2007) 1649–1656.
[6] J. Liquier, A. Akhebat, E. Taillandier, F. Ceolin, T. Huynh Dinh, J. Igolen. Spectrochim. Acta. 47 (1991) 177–186.
[7] H. Malonga, H. Arakawa, J.F. Neault, H.A. Tajmir-Riahi. DNA Cell Biol. 25 (2006) 393–398.
[8] Ornella Preisner,Joao Almeida Lopes, Raquel Guiomar, Jorge Machado, C. José Menezes. Anal. Bioanal. Chem. 387 (2007) 1739–1748.
[9] A. Pichler, S. Rudisser, R.H. Winger, K.R Liedl, A.Hallbrucker, E.Mayer. Chem. Phys. 258 (2000) 391–404.
[10] F.V. Subacha, J. Liquierb, Е.S. Gromova. Russ. J. Gen. Chem. 78 (2008) 1103–1109.
[11] T. Amber Krummel, T. Martin Zanni. J. Phys. Chem. B. 112 (2008) 1336-1338.
[12] Canh Le-Tien, Roxanne Lafortune, Francois Shareck, Monique Lacroix. Talanta71 (2007)1969–1975.
[13] H.A. Tajmir-Riahi, C.N. soukpoé-Kossi, D. Joly. Spectroscopy 23 (2009)81–101.
[14] Martina Lindqvist, Astrid GraÈslund. J. Mol. Biol. 314 (2001) 423-432.
[15] L.M. Nie, W.Q. Zhang, M. Xiao, J.L. Luo, K. Bao, J.K. Chen, B. Li. J. Phytopathology. 155 (2007) 364–367.
[16] Venkata Radhakrishna Kondepati, Michael Keese, H. Michael Heise, Juergen Backhaus. Vib. Spectrosc. 40 (2006) 33–39.
[17] Annette Naumann, Gregor Hein, Rolf Rauber. Field Crops Res. 119 (2010) 78–84.
[18] Koji Emura, Shinsuke Yamanaka, Hiroko Isoda, N. Kazuo Watanabe. Breeding Sci. 56 (2006) 399-403.
[19] D. haram Singh, Paramvir Singh Ahuja. Genome Res. 49 (2006) 91–96.
[20] T. Balasaravanana, P.K. Pius, R. Raj Kumar, N. Muraleedharan, A.K. Shasany. Plant Sci. 165 (2003) 365–372.
[21] Tapan Kumar Mondal.Euphytica128 (2002) 307–315.
[22] S.Q. Que, F. Zhang. J. Anhui Agri. Sci. 36 (2008) 902 - 905.
[23] J.B. Shen, H.F. Lu, Q.F. Peng, J.F. Zheng, Y.M. Tian. J. Syst. Evolution. 46 (2008) 194–204.
[24] Gunjan Tyagi, K. Deepak Jangir, Parul Singh, Ranjana Mehrotra. DNA Cell Biol.00 (2010) 1–7.
[25] Martina Banyay, Astrid Gra slund. J. Mol. Biol. 324 (2002) 667–676.
[26] D. Dohy, M.Ghomi, E. Taillandier. J. Biomol. Struct. Dyn. 6 (1989)741–754.
[27] E. Taillandier , J. Liquier. Methods Enzymol. 211 (1992) 307–335.
[28] R. Letellier, M. Ghomi, E. Taillandier. J. Biomol. Struct. Dyn. 6 (1989).
[29] W.K. Olson, A.A. Gorin, X.J. Lu, L.M. Hock, V.B. Zhurkin. Proc. Natl Acad. Sci. USA. 95 (1998) 11163-11168.
[30] S. Jones, P. van Heyningen, H.M. Berma, J.M. Thornton. J. Mol. Biol. 287 (1999) 877-896.
[31] A.A. Ouameur, H.A. Tajmir-Riahi. J. Biol. Chem. 279 (2004) 42041–42053.
[32] S. Alex, P. Dupuis. Inorg. Chim. Acta.157 (1986) 271–281.
[33] M. Loprete, K.A. Hartman. Biochemistry. 32 (1993) 4077–4082.
[34] E. Taillandier, W.L. Peticolas, S. Adam, T. Huynh- Dinh, J. Igolen. Spectrochim. Acta part A. 46 (1990) 107–112.
[35] J. Liquier, E. Taillandier. IR Spectrosc. Biomol. 35 (1996) 131–158.
[36] R. Letellier, M. Ghomi, E. Taillandier. J. Biomol. Struct. Dyn3 (1986)671–687.
[37] R. Ahmad, H. Arakawa, H.A. Tajmir-Riahi. J. Bio. phys. 84 (2003) 2460–2466.
Web design by Tribal Systems
