Pseudopollen in the Genus Camellia, its Definition, Morphology and Status
Wang Zhonglang* 1, Noriyuki Fujishita2, Toshiaki Shiuchi3, Masashi Nakata3
1Kunming Institute of Botany, Chinese Academy of Sciences, Kunming 650204, China
2Osaka University, Osaka, 589-0022, Japan
3Botanic Gardens of Toyama, Toyama 939-2713, Japan
* Author for correspondence, e-mail: wang@mail.kib.ac.cn
Introduction
The genus Camellia is the largest and economically the most important one in the family Theaceae. The species of Camellia are mainly distributed in southeastern and eastern Asia, especially in China, which is the center of species diversity, holding more than 80% of the total species in the genus (Min 2000, Gao et al. 2005). The most economically important species in Camellia is the beverage tea (C. sinensis). Currently more than 40 countries commercially produce the tea, and about 3.6 million tons of tea leaves are annually produced (Chen et al. 2007). Tea leaves have been reported to have more than 700 chemical constituents, of which catechins, flavonoids, vitamins (C, E, K), amino acids, caffeine, and polysaccharides are very important for human health, benefitting the treatments for many diseases such as cancer, diabetes, obesity, and many cardiovascular ailments (Khan and Mukhtar 2007, Murase et al. 2002). Camellia is also important in horticulture. Many plants in the genus are grown as woody ornamentals worldwide, especially those from the species of C. japonica, C. reticulata, C. sasanqua. Recently a group of yellow-flowering camellias as well as the ever-blooming species, C. azalea, are popularly cultivated in many countries for ornamental purpose. The third important use of Camellia is the oil industry. Many species of Camellia are used for extracting oil from seeds, especially the species C. oleifera that has been historically cultivated for oil in China for more than 2,000 years. The oil is widely used in China for cooking and cosmetic purposes (Min 2000, Zhang et al. 2007). In China, currently more than 3 million ha are used for camellia oil production, and ca. 1 million tons of seeds are harvested each year, which yield nearly 260,000 tons of oil. According to the business plan of the national camellia oil industry, in 2020, the area will reach to 4.7 million ha, and will produce 2.5 million tons of camellia oil (SFAC, 2009).
Pollen grain is the male sex cell of a flowering plant, very important for breeding new cultivars. Pollen grains are protected by thin, glassy, plastic covers, which are highly resistant to decay, which also have very stable characteristics for classification. Actually, these tiny, fragile-looking granules are more durable than any other part of a tree (Hesse et al. 2008). It has been reported as an important characteristic in the taxonomy of the family Theaceae (Wei et al. 1992, Zavada and Wei 1993, Wei 1997, Ao et al. 2002). In 1997, two researches discovered almost simultaneously a striking feature that some genera of the family Theaceae possessed pseudopollen grains (PPG) by different methods (Tsou 1997, Fujishita 1997). The PPG were then used as one of important characters to classify the genera in the family of Theaceae according to the results obtained by Tsou in 1997(Tsou 1998, Hesse 1999, Prince and Parks 2001, Li et al 2003, Wang et al. 2006, Vijayan et al. 2009).
Besides the plants in Theaceae, many others were also found to possess PPGs. Kawakubo(1990) found PPG from three species in Callicarpa that showed special function on ‘cryptic dioecy’, as the female flowers produced non-germinating pollen (PPG). It has also been testified later that the pseudopollen grains were served as rewards for pollinators (Kawakubo 2007). Sterile pollen grains of cryptic female flowers have also been reported for some species in Solanum (Knapp et al. 1991) as well as in Rosa (Kevan et al. 1990). The PPG have been reported in many species of the family Ochidaceae. For example, Davies has published a series of papers on PPG in the genera Maxillaria, Erica, Polystachya of Ochindaceae to elucidate the structure, development, functions, constituents as well as distribution diversity (Davies 1998, Davies et al. 2000, 2002, 2003, 2004). But in the genus Camellia the definition and distribution of PPG was so far not clear yet, and there was a lack of knowledge on the constituents and structure.
The aim of present paper is to examine the morphology and ranges of pseudopollen morphology in 50 species as well as six interspecific hybrids, to give a clear definition of a pseudopollen grain in the genus Camellia, which has many economically important species as shown in the above. This could promote the understanding of the pollination system and classification of the species in the genus Camellia.
Material and methods
Pollen was removed from the camellia plants cultivated so far in Japan (Table 1). Pollen grains were released on a glass slide by gently squashing anthers with fine forceps in a drop of idodine-potassium iodide staining solution (1 g I2 and 2g KI dissolved in 300 ml distilled water). Anther debris was removed carefully and a cover glass was applied over the staining solution before examination under a microscope (Nikon Eclipse E400, Japan). During observation, a small drop of Methanesulfonic Acid (CH3SO3H) was added for confirming the existence of pseudopollen grains.
Dry pollen grains for SEM were attached to aluminium stubs using double-sided sticky carbon tabs (DTM 9101), then coated with gold by sputtering device (JEOL JFC-1100E 10N, Japan) at 10 mA for 4 min, examined and photos taken using a Scanning Electron Microscope (JEOL JSM-T20, Japan) at an accelerating voltage of 10 kV.
Results and discussion
The morphology of pseudopollen grain in genus Camellia
The definition of pseudopollen is very important for plants in different families, it seems necessary to be specified in different taxa from the view of former studies (Kawakubo1990, Kevan et al. 1990, Knapp et al. 1991, DaviesandTurner2004b), though all pseudopollen grains were also derived from anthers. The pseudopollen in the family Orchidaceae, for instance, was briefly defined as “a whitish, mealy material produced upon the labella of a number of orchid species as labellar hairs either become detached or fragment. Since individual hair cells are rich in protein and starch, its function possibly serves as a reward for visiting insects” (Davies and Turner, 2004b). What is a pseudopollen grain in the genus Camellia, and how to tell the difference from the normal pollen grains? A brief comparison was shown as follows:
In general, the normal pollen grains in the genus Camellia were suboblate, subspheroidal to subprolate in shape, usually 3-colporate. Pollen size varies from species to species, usually 35-45(-55) mm x37-48(-77)mm, the exine sculpture could divided into six types, useful for the taxonomic classification of the genus Camellia (Wei et al. 1992, Ao et al. 2002).
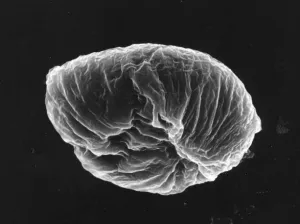
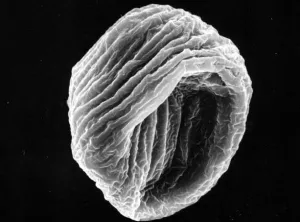
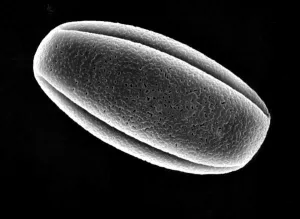
The pseudopollen grains in the genus Camellia, on the other hand, we found 1) they had much more variable shapes, some were regular shapes similar to normal pollen grains, but most of them were in irregular shape by being deflective, sunk or even cupped (Fig.1: A, B, C, D, F ). 2) No aperture at all was found in the exine of PPG (Fig.1: F ). It is well known that apertures are key characteristics of pollen grains with systematic importance in angiosperms, functioning as sites for pollen tube exit, water uptake, transfer of recognition substances and accommodation of volume changes (Hesse, M et al. 2009 ). Thus, the PPG could not germinate after pollination. 3) PPGs are generally similar in size to the normal pollen grains, but with a much greater range of variation (20-80 mm). If the normal pollen grains were big in size, the PPG were also big, and vice versa (Fig.2: C, D).
 |
 |
|
A PPG of C. saluenensis |
B PPG of C. hongkongensis |
 |
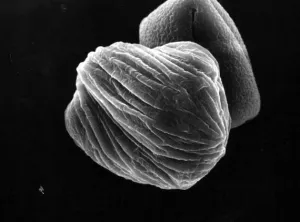 |
| C PPG of C. pitadii | D PPG of C. yunnanensis |
 |
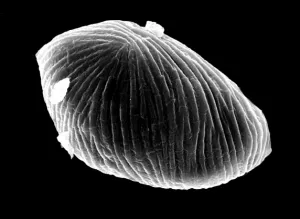 |
| E normal pollen grains of C. reticulata | F PPG of C. reticulata |
Fig. 1 PPG morphology under SEM
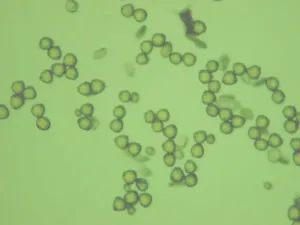 |
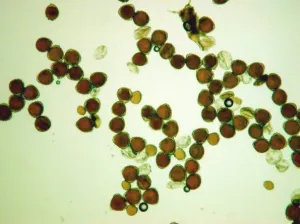 |
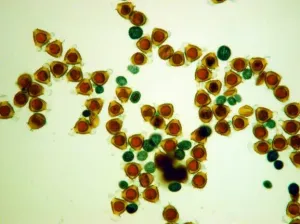 |
 |
 |
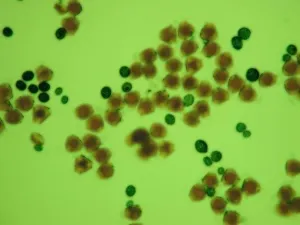 |
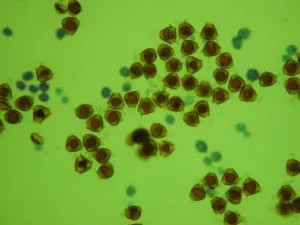
Fig. 2 Comparison of PPG to normal pollen grain under LM
G-I Same field of vision in same plate of wild Camellia reticulata showing the changes in color.
The field has only slightly difference, four pollens in I as shown by pink arrow moved from up-right in G and H after adding strong acid.
G. Directly in distilled water. It is very difficult to distinguish PPG from normal pollen grains.
H. Stained in idodine-potassium iodide staining solution. The normal pollen grains are in orange color, while PPG in very pale color.
I. Immediately after adding methanesulfonic acid, the color of PPG change to blue, very easy to distinguish.
J In the same stage of C but different species, namely Camellia yunnanensis.
K-L Same field of vision but in different focus to show the normal pollen grains are in the upper layer in K in the focus, but PPG in the bottom layer in L in the focus.
4) The exine of PPG is so different from that of normal pollen grain under SEM, which composed of about 30 to 50 parallel ribs to form a Scallop shell-like structure(Fig.1: F). 5) PPG was heavier in gravity than normal pollen, if put both of them in water, the PPG is at the bottom layer, while the normal pollen in the upper layer(Fig.2: K,L). 6) If methanesulfonic acid is added immediately after the staining solution of idodine-potassium iodide, the color of PPG will change quickly into blue color while the normal pollen grains are still in orange color (Fig. 2: G, H, I). So it is very easy to distinguish PPG from normal pollen grains under light microscope by blue color (Fig.1: J). It is possibly because the wall of PPG is easily dissolved in strong acid, so the starch inside reacts with iodide in the staining solution, leading to the appearance of the blue color. The wall of normal pollen is highly resistant to decay, which is more durable than that of pseudopollen grain.
The distribution of pseudopollen grains
Tsou (1995) found that all three genera, Adinandra, Cleyera, and Eurya of Ternstroemioideae of Theaceae did not possess PPG. On the contrary, all ten genera examined of the Camellioideae of Theaceae produced PPGs, and could be divided into three patterns by the secondary wall thickening of PPGs (Tsou 1996, 1997): the thickenings found in Camellia, Godonia, Hartia, Pyrenaria, Sinopyrenaria and Tutcheria are composed of about 30 to 40 parallel ribs. The thickened walls of Franklinia and Schima resemble pantoporate hollow balls, whereas Apterosperma and Stewartia show an intermediate pattern. Tsou (1997) describes this feature as ‘the only autapomorphy so far determined’ for this subfamily. Iqbal et al (2002) also reported that two Gordonia species in same subfamily Camellioideae produced pseudopollen.
The genus Camellia consists of more than 200 species, but the conclusion Tsou came to in this genus was only from two species, namely Camellia hengchunensis and C. tenuifolia. A question arises, do all the other species in the same genus also produce PPG? As we have a very easy method to test for the existence of PPG, a survey was carried out to check the pollens of all the species we could obtain. The results showed that many species in the genus Camellia did possess pseudopollen grains (called PPG-forming species), but there remains some species that have not been found to produce pseudopollen grains (non PPG-forming species).
From the previous study of Fujishita 2005, amongst 50 wild species studied, 30 species were PPG-forming species while 20 are non-PPG-forming species. Most species in Subgenus Protocamellia and Subgenus Camellia are PPG-forming species, except three species in Sect. Camellia, namely C. chekiangoleosa, C. japonica and C. hozanensis. Vijaya et al. (2009) analyzed ITS sequences of 112 species of Camellia, and found 25 species from the Section Camellia were arranged in three clades, while all species in the sections Theopsis, Thea and Furfuraceae were arranged in the corresponding sections respectively. The southern and SE species in China as well as in Japan, namely C. japonica, C. chekiangoleosa, C. rusticana et al. are grouped into one clade, which were easy to distinguish them by the persistence of bracteloes and sepals up to the fruiting stage. In this study we found they belong to non PPG-forming species. The SW China species, i.e. C. reticulata, C. pitardii grouped into one clade, are PPG-forming species, which are largely restricted to higher elevated regions in Sichuan, Guizhou and Yunnan provinces. The PPG data supported the Section Camellia is polyphyletic made by ITS molecular data. Almost all the species in Subgenera Thea and Metacamellia are non PPG-forming species, except Camellia forrestii, which is mainly distributed in SW China.
Tsou (1997, 1998) suggested pseudopollen was the unique to members in Camellioidea of Theaceae, but from our research, not all members in the genus Camellia produced pseudopollen. This is not a suitable characteristic to classify the genera in Theaceae, although many authors had already used (Tsou 1998, Hesse 1999, Prince and Parks 2001, Li et al 2003, Wang et al. 2006, Vijayan et al. 2009).
Heredity of PPG in F1 hybrids
From Table 2, we could obtain the result that the gene for forming PPG is dominant to the non forming gene If one parent is PPG-forming species, no matter whether it was a male or female parent, the F1 generation would also produce PPG. If both parents were non PPG-forming species, the F1 were the same, so hybridization could not increase the opportunity to possess PPG, but could decrease the ratio of normal pollen grains, to produce more abnormal pollen grains, especially the empty pollen grains.
Table 2 The detailed data of different kinds of pollen in F1 hybrids*
|
No. |
Scientific Name |
normal pollen (NP) |
uneven pollen
|
empty pollen
|
starch pollen
|
big pollen
|
small pollen |
PPG |
NP ratio (%) |
PPG ratio (%) |
|
1 |
C. japonica 'Oniji' (a triploid 3x) |
177 |
38 |
235 |
53 |
61 |
33 |
0 |
31.4 |
0.0 |
|
2 |
C. Minato-no-Akebono' (C.lutchuensis x C.japonica) |
77 |
43 |
332 |
220 |
10 |
1 |
0 |
11.3 |
0.0 |
|
3 |
C. 'Shokou' (C.japonica x C.chrysantha) |
32 |
82 |
337 |
184 |
0 |
2 |
0 |
5.0 |
0.0 |
|
4 |
C. 'Brian' (C.saluenensis x C.reticulata) |
317 |
5 |
179 |
64 |
1 |
3 |
40 |
52.3 |
6.6 |
|
5 |
C. 'Promises' (C.reticulata x C.granthamiana) |
537 |
15 |
71 |
11 |
3 |
32 |
80 |
74.1 |
11.0 |
|
6 |
C. 'Fallen Angel' (C.japonica x C.granthamiana) |
0 |
30 |
433 |
0 |
49 |
0 |
84 |
0.0 |
14.1 |
|
7 |
C. ‘Chachahime' (C.rusticana x C.sinensis) |
266 |
35 |
298 |
8 |
35 |
201 |
0 |
41.4 |
0.0 |
|
8 |
C. 'Patricia Ann' ('Berenice Boddy' x 'Mrs D.W Davis') |
333 |
38 |
211 |
9 |
39 |
46 |
0 |
52.8 |
0.0 |
* please check the definition of normal pollen, uneven, empty, starch, big or small pollen grains in Fujishita (2000).
References
Fujishita N. 1997. The diversity of pollen morphology and physiology, II: view from the materials of Camellia and some other genera in the family Theaceae, Abstracts of the papers presented at the 38th annual meeting of the Palynological Society of Japan. 19 (In Japanese)
Fujishita N. 2000. Abnormal Pollen—reminiscences and the latest results (Ⅰ). Jpn. J. Palynol. 46(2):163-178. (in Japanese)
Fujishita N. 2001a. Abnormal Pollen—reminiscences and the latest results (Ⅱ). Jpn. J. Palynol. 47(1):75-93. (in Japanese)
Fujishita N. 2001b. Abnormal Pollen—reminiscences and the latest results (Ⅲ). Jpn. J. Palynol. 47(2):137-154. (in Japanese)
Fujishita N. 2002a. Abnormal Pollen—reminiscences and the latest results (Ⅳ). Jpn. J. Palynol. 48(1):41-54. (in Japanese)
Fujishita N. 2002b. Abnormal Pollen—reminiscences and the latest results (Ⅴ). Jpn. J. Palynol. 48(2):129-152. (in Japanese)
Fujishita N. 2003. Interspecies hybridization method of Camellia by activating the specification of pollen grains. Tsubaki, 42:28-38 (in Japanese)
Hertog, M. G. L., Feskens E. J. M., Hollman P. C. H. et al. 1993. Dietary antioxidant flavonoids and risk of coronary heart disease: the Zutphen Elderly Study. Lancet 342: 1007–1011
Hesse,M., Halbritter,H.,Zetter, R. et al., 2009. Pollen terminology: an illustrated handbook . Springer Wien, New York. pp264
Huang T.C. 1972. Pollen Flora of Taiwan, Nat.Taiwan Univ. Bot. Dep. Press, Taipei, pp.273, pls 177.
IMAI, K. AND K. NAKACHI. 1995. Cross sectional study of effects of drinking green tea on cardiovascular and liver diseases. Brit. Med. J.310: 693–696
Iqbal M.C.M., Wijesekara K.B. 2002. Cells of the connective tissue differentiate and migrate into pollen sacs. Naturwissenschaften 89:39-42.
Johns C., Lu M.Q., Lyznik A., and Mackenzie S. 1992. A mitochondrial DNA sequence is associated with abnormal pollen development in cytoplasmic male sterile bean plants. The Plant Cell, 4:435-449
Kataoka H., Morita Y. and Miyoshi, 2001. Pollen morphology by means of scanning electron microscope 14. Theaceae. Jpn. J. Palynol. 47(1):1-12 (in Japanese with English abstract)
KATIYAR, S. K., R. AGARWAL, AND H. MUKHTAR. 1993. Protection against malignant conversion of chemically induced benign skin papillomas to squamous cell carcinomas in SENCAR mice by a polyphenolic fraction isolated from green tea. Cancer Res. 53: 5409–5412.
Kawakubo N. 1990. Dioecism of the genus Callicarpa (Verbenaceae) in the Bonin (Ogasawara) Islands. Bot. Mag. Tokyo, 103:57-66
Kawakubo N. 1998. Evolution of crytic dioecy in Callicarpa (Verbenaceae) on the Bonin Islands. Stuessy T.F. and Ono M. (eds). Evolution and Speciation of Island Plants. Cambridge University Press, Cambridge. 155-168
Kawakubo N. 2007. Evolution of cryptic dioecy in Callicarpa (Verbanaceae) on the Bonin Islands. In: Stuessy T.F. and Ono M.(eds). Evolution and Speciation of Island Plants. Cambridge University Press 155-168.
Kevan P.G., Clark E.A. and Thonas V.G. 1990. Pollination: a crucial ecological and mutualistic link in agroforestry and sustainable agriculture. Proc. Ent. Soc. Ont. 121:43-48
Knapp, S., V. Persson & S. Blackmore. 1998. Pollen morphology and functional dioecy in Solanum (Solanaceae) Plant Sys. Evol. 210: 113-139.
Prince, L. M. and Parks C.R., 2001. Phylogenetic relationships of Theaceae inferred from chloroplast DNA sequence data. Amer. J. Bot. 2001;88:2309-2320
Prince, LM. 2007. A brief review of 20th century nomenclature for genera and tribes of Theaceae. Aliso 24: 105–121.
SFAC (State Forestry Administration of P.R. China). 2009. The development roadmap of national camellia oil industry from 2009 to 2020. (In Chinese ) http://www.sdpc.gov.cn/zcfb/zcfbtz/2009tz/ W020091105649468359531.pdf
Tsou C. H. 1997. Embryology of Theaceae - Anther and ovule development of Camellia, Franklinia and Schima. Amer. J. Bot. 84: 369-381
Tsou C. H. 1998. Early floral development of Camellioideae (Theaceae). Amer. J. Bot. 85:1531-1547
Tsou, C. H. 1995. Embryology of Theaceae -- anther and ovule development of Adinandra, Cleyera, and Eurya. J. Plant Res. 108: 77–86.
Tsou, C. H. 1996. The pseudopollen of the Camellioideae :its development and systematic significance. Amer. J. Bot. 83(Supplement):198-199 (Abstract)
Vijayan K., Zhang W. J. and Tsou C.H. 2009. Molecular taxonomy of Camellia (Theaceae) inferred from nrITS sequences. Amer. J. Bot. 96, 1348-1360.
Wang Y. H., He H., Min T. L., Zhou L. H. and Fritsch P. W., 2006. The phylogenetic position of Apterosperma (Theaceae) based on morphological and karyotype characters. Plant Sys. Evol., 260 (1):39-52
Wei Z. X. ,1997. Pollen ultrastructure of Theaceae and its systematic significance. Act. Bot. Yunn. 9: 143–153.
Wei Z. X., Zavada M. S. and Ming T. L. 1992. Pollen morphology of Camellia (Theaceae) and its taxonomic significance. Act. Bot. Yunn. 14. (3): 275-282
Zavada, M. S. and Wei, Z.X. 1993. A Contribution to the Pollen Morphology of Camellia (Theaceae), Grana, 32: 4, 233-242
Web design by Tribal Systems
