Fungal transmission of a color-breaking virus in Tama-no-ura camellia in Japan
Chuji Hiruki*
Department of Agricultural, Food and Nutritional Science, University of Alberta,Edmonton, Alberta T6G 2P5, Canada
* Author for correspondence: chujihiruki@aol.com
Introduction
Of the many species of Camellia family, Camellia japonica, indigenous to Japan and the southeastern Asia, was grown as garden specimens as early as the seventh century in Japan (Nakamura, 1975). At the present time, it is grown in many areas of moderate climate in the world as valuable ornamentals in gardens and greenhouses. This species has numerous cultivars in a varying range of colors of reds, pinks, mauvish reds, creams, whites and many bi-colors. It is not uncommon for collectors to have more than 1,000 different cultivars in their gardens. C. japonica ‘Tama-no-ura’ (Fig. 1), (Hiruki, 2008), a bi-color picotee mutant, was found growing as a mature tree, about 200 year-old, in a deep forest for the first time in 1947 by a charcoal worker in Tamanoura Town, Goto islands, Japan.

Fig. 1 A healthy ‘Tama-no-ura’ camellia
As soon as it was introduced at a camellia show in Nagasaki in 1973, it was instantly famous as it was believed to be very similar to one described in an ancient scroll in Japan and thought to be extinct. ‘Tama-no-ura’ has been accepted by many enthusiasts around the world for not only by its own outstanding beauty but also for its excellent genetic trait as a dominant gene source.
The color-breaking of ‘Tama-no-ura’ was found to be caused by a virus causing a green mottle and spots in the leaf tissue of the infected camellia plants. The mode of soil transmission was established to be vectored by an Olpidium sp. for the first time in this study.
Materials and Methods
Plants
A camellia seedling suspected of being infected with a virus was obtained in 2004. The potted plant of ´Tama-no-ura´ showed an irregular wavy pattern in the white edge of petals (Fig. 2) and pot soil in which the plant was growing was used in the present study.

Fig. 2 ’Tama-no-ura’ camellia with wavy patterns on the petal suggesting virus infection
Growth conditions
Unless otherwise stated, camellia seedlings were grown in 17 cm-diameterplastic pots containing a commercial premixed soil (Registered name: Sunshine mix LA4, peat/perlite/vermiculite/sand) from Sun Gro Horticulture Canada Ltd., Seba Beach, AB T0B 2B0, Canada.
Greenhouse temperature was maintained at 21±1ºC during day and 18ºC at night. Light period was 16 hrs with a light intensity of 400 mv provided with C5F54T5/835/color HO Highoutact band lamps. Fertilizer (15/30/15) was provided biweekly at a concentration of 200 ppm.
Scanning Electron microscopy
Methods employed were the same as published previously (Hiruki and Alderson, 1976).
Results
Detection and isolation of viruliferous Olpidium sp. from infested camellia soil
In preliminary tests including several species of plants, tobacco (Nicotiana tabacum) was found to be most susceptible to Olpidium. Young tobacco seedlings were used as a trapping plant for the detection and isolation of Olpidium.
Zoosporangia and zoospores were found in the root tissues of 3 repeated seedings of tobacco for trapping. Resting spores typical for Olpidium were found at a late stage of infection (Fig. 3).
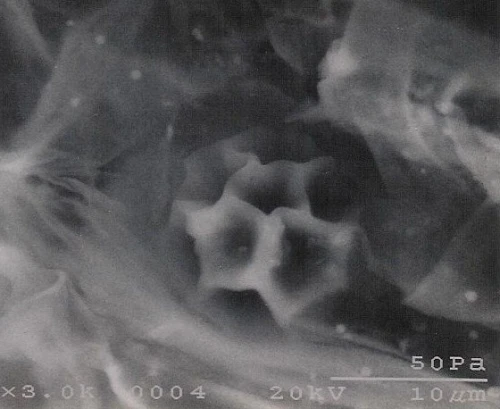
Fig.3. A resting spore of Olpisium sp.
Inoculation of camellia seedlings with viruliferous Olpidium
After establishing a culture of viruliferous Olpidium in tobacco, Olpidium-infected roots were air-dried and pulverized in a mortar with a rubber stopper. About one gram of Olpidium-containing root powder was mixed with 9 grams of autoclaved soil. The mixture was spread thinly over pot soil in each pot and camellia seeds were placed in the mixture. About one month after the seeding, tobacco seeds were sowed in the same pot for Olpidium detection as it was found easier to find Olpidium in young tobacco roots, although Olpidium was found in camellia roots as well (Fig. 4).

Fig.4 Olpidium infection in camellia tree was detected easily by using young tobacco seedlings as a trap plant.
Detection of Olpidium from virus infected camellia plants
Observation of green mottle symptom development and the presence of Olpidium in the camellia roots and pot soil were continued for 5 years using 5 camellia seedlings after inoculation of germinating camellia seedlings in 2006 (Fig.5).
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Fig. 5. Green mottle symptoms in the inoculated camellia seedlings
The symptoms of green mottle developed 3 to 5 months after inoculation. The typical green spots and mottle developed each year as new leaves emerged. In 2009 the first floral buds were formed. Color-breaking of typical picotee in Tama-no-ura was observed in the camellia plants artificially inoculated by mean of Olpidium in this study (Fig. 6).

Fig. 6. Color-breaking of ‘Tama-no-ura’ after artificial inoculation in the greenhouse.
Discovery of naturally infected Tama-no-ura camellia showing color-breaking in the field
In 2006, a Tama-no-ura camellia about 10-years old growing in the field was found showing color-breaking (Fig. 7, 8). Leaf symptoms in the infected plant were not very distinct as it was exposed to sun in the field, but flowers displayed severe color-breaking of various degrees. This tree and the original potted plant obtained in 2004 originated from a same supplier.
|
|
|
| Fig.7 Color-breaking of ‘Tama-no-ura’ found on the naturally infected camellia plant in the field | Fig.8 Naturally virus-infected plant of ‘Tama-no-ura’ camellia in the field |
Discussion
First case in definitive reduction of the aesthetic value of a camellia cultivar
There are numerous cases of color-breaking in camellias and some of them are prize winners in camellia flower shows, like ‘Donckelaeri’ (=’Masayoshi’). In some cases, the introduction of virus by means of grafting was reported previously (Plakidas, 1948, 1952, 1954; Tourje, 1950) and it was suggested that grafting was a useful means of producing attractive new cultivars (Plakidas, 1948). However, in the case of ‘Tama-no-ura’, virus infection is definitely detrimental to its aesthetic value and results in a drastic reduction in the saleability of the seedlings in the market. Thus, virus infection of ‘Tama-no-ura’ should be avoided by all means.
First case of fungus transmission of a camellia virus
Sap transmission of virus is very common in many virus infections in plants. However, camellia variegation (=yellow mottle) virus could not be transmitted by sap (Hiruki, 1985). In this paper fungal (Olpidium sp.) transmission of the virus in ‘Tama-no-ura’ has been established for the first time、although the result from a previous electron microscopy study (Hiruki, 1985) suggested that camellia variegation virus morphologically belongs to a varicosa virus that is transmitted by Olpidium (Hiruki, 1996).
Perspectives in the future studies
There are many cases of color-breaking in camellia cultivars that are suspected to be caused by a virus or viruses (Milbrath et al.,1946). However, there are no data to relate each case to establish relationships among presumptive viruses, although a suggestion was made for the presence of strains of a camellia virus (Plakidas, 1962). Detailed molecular studies are expected to shed light on the classification of camellia viruses in the near future.
Acknowledgements
The author would like to thank the International Camellia Society for a generous financial support of this project with the Otomo Fund.
References
Hiruki, C. 1985. A preliminary study on infectious variegation of camellia. Acta Horticulturae 164: 55-62.
Hiruki, C. 1996. Camellia yellow mottle(?) varicosa virus. In: Viruses of Plants, Descriptions and Lists from the VIDE Database. pp. 285-286. (Editors.) Brunt, A., Crabtree, K., Dallwitz, M., Gibbs, A., Watson, L. CAB International, Oxon, UK.
Hiruki, C. 2008. Tama-no-ura camellia: the current situation, history, and future possibilities. International Camellia Journal 2008, 66-70.
Hiruki, C. and P. G. Alderson. 1976. Morphology and distribution of resting sporangia of Olpidium brassicae. Canadaian Journal of Botany 54:2820-2826.
Milbrath, J.A. and McWorter, F. P. 1946. Yellow mottle leaf, a virus disease of camellia. American Camellia Yearbook 1946: 51-53.
Nakamura, T. 1975. [Camellia and Sasanqua] (in Japanese). pp. 176. Seibundo-Shinkosha Publisher, Tokyo.
Plakidas, A. C. 1948. Possibility of utilizing virus infection as a means of producing new varieties of camellias. American Camellia Yearbook 1948: 107-109.
Plakidas, A. C. 1952. Transmission of leaf and flower variegation in Camellia japonica. Louisiana Agricultural. Experiment Station Annual Report 1950-51: 115-116.
Plakidas, A. C. 1954. Transmission of leaf and flower variegation of camellia by grafting. Phytopathology 44:14-18.
Plakidas, A. C. 1962. Strains of the color-breaking virus of camellia. Phytopathology 52: 77-79.
Tourje, E. C. 1950. Virus transmission through grafting. Camellia Research (Southern California Camellia Society) 68-71.
Web design by Tribal Systems




















