Ultrastructure of Cells in Formation and Release of Volatile Compounds in an Aromatic Camellia Cultivar
Fan Zhengqi*, Li Jiyuan, Li Xinlei, Yin Hengfu and Zhou Xingwen
Research Institute of Subtropical Forestry, CAF, Fuyang, Zhejiang 311400, China
* Author for correspondence, e-mail: fzq_76@126.com
Introduction
Fragrance in camellias is one of the most important horticultural characters of the famous camellia varieties. Only a few varieties, perhaps ten, emit scents although thousands of varieties are grown in the world. Most camellias have no fragrance. At present, breeding of camellia varieties with peculiar colors and fragrance are key goals for camellia breeders. The compounds related to floral scents have been determined in Camellia sinensis (Gu et al, 2008), Camellia renshanxiangiae (Song et al, 2009) and Camellia grijsii (Fan et al, 2006), Camellia japonica ‘Kramer’s Supreme’ and Camellia japonica ‘Scentsation’ (Fan et al, 2006). The fragrant varieties contain the main volatile compositions such as terpenes, esters, ancohols, aldehydes and acids, and there were some differences among varieties and species. Camellia japonica ‘Kramer’s Supreme’ is a famous and rare aromatic variety of camellia, emitting a strong fragrance with main components being linalool, benzyl benzoate, methyl salicylate, and so on.
The volatile compounds related to floral scent are synthesized in cells. The ultrastructure of floral organs were previously observed in Magnolia denudata Desr. (Cai et al, 2001), Erysimum cheiri (Weston, et al, 1999), Osmanthus fragrans (Chang, et al, 2007), Chamelaucium uncinatum (O’Brien, et al, 1996) and Dianthus caryophyllus L. (Smith et al, 1992), but not yet in Camellia. This paper focuses on the ultrastructure of cells, and the accumulation of volatile compounds in Camellia japonica ‘Kramer’s Supreme’ flowers. The relation between ultrastructure and volatile oils will be discussed.
Materials and Methods
Plant materials
Camellia japonica ‘Kramer’s Supreme’, grown in the Research Institute of Subtropical Forestry, CAF, Fuyang, Zhejiang, China, was collected for this study. The sample of petals and filaments were collected in March and April.
Transmission Electron Microscopy (TEM)
Tissue was fixed in 3% (v/v) glutaraldedhyde overnight at 4℃, and washed three times with 0.1 M Na-K-phosphate buffer (pH 7.0). Tissue were then postfixed in 1% OsO4 for 1-2h, washed three times with the same buffer, dehydrated in a graded series of ethanol (50%, 70%, 80%, 90%, 95% and 100%). The samples were treated with a solution of acetone and embedding medium (V/V=1/1) for 1h, (V/V=3/1) for 3h, prior to the penetrated sample being embedded in embedding medium overnight.
Sections 70-90 nm thick obtained at Reichert superthin slicer, were stained with lead citrate and 50% ethanol solution of uranyl acetate for 15 min, respectively, and then obverved with a JEOL JEM-1200(1230) electron microscope.
Results
Ultrastructure in cells of floral organs
The ultrastructure of cells in petals (Figure 1A) and filaments (Figure 1B) of Camellia japonica ‘Kramer’s Supreme’ were observed. The two kinds of cells in floral organs both contained some subcellular structures such as nucleus, plasmid, mitochondria, endoplasmic reticulum and golgi apparatus (Figure 1C). The large vacuole and little nucleus were found in most cells of petals, while thick cytoplasm, big nucleus and small vacuole were contained in a few cells in petals and filaments. The cells in filament had more mitochondria and plasmids, with developed endoplasmic reticulum; cells in petals contained the deep stained grains, which were probably pigments (Figure 1D). Plasma membranes were different from filament ones. The plasmids in cells were round or elliptical.
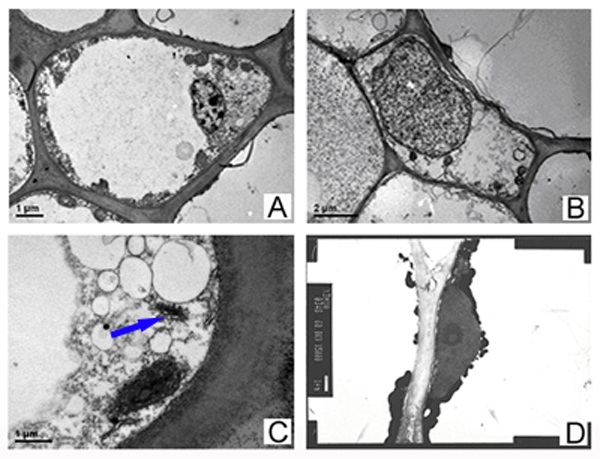 Figure 1 Ultrastructure of cells in Camellia japonica ‘Kramer’s Supreme’ flowers
Figure 1 Ultrastructure of cells in Camellia japonica ‘Kramer’s Supreme’ flowers
A. Cell in petals; B. Cell in filaments;
C. Chloroplast of cell in filaments; D. Golgi body (arrowed)
The ultrastructure of volatile compounds during formation and emission
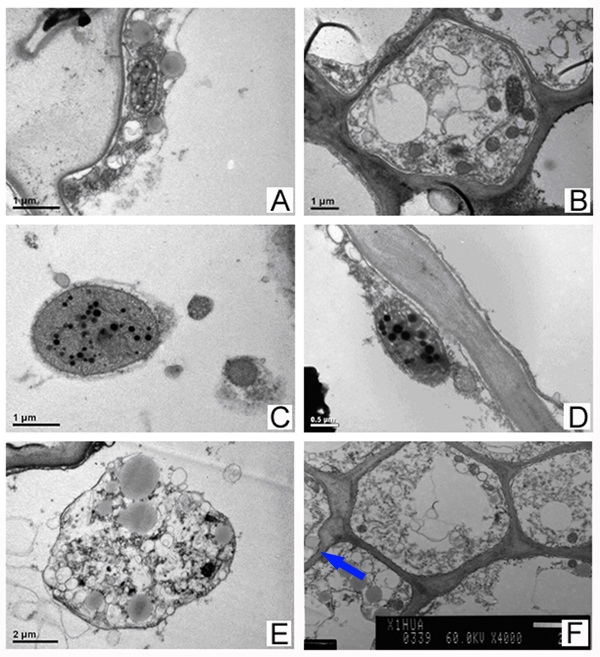
Figure 2 Formation and release in volatile oil
A. Grey oil drop; B. Plastids in cell;
C.Oil produced in plastid; D. Larger oil in plastid;
E. Oil packed with membrane while plastid disintegrating; F.Membrane of oil drop merged into cell membrane (arrowed).
A lot of grey osmiophilic granules were found to be packed with membrane (Figure 2A), which presented in some cells petal and filament that contained many plasmids (Figure 2B), possibly these were sources of volatile oils of the aromatic camellia. Some previous studies indicated that volatile oils are synthesized in plasmids, which is consistent with our results. The sizes and color of osmiophilic granules were different in plasmids (Figure 2C), and the volatile composition probably synthesized here. The oil drops in plasmid were not packed with membrane in plasmids (Figure 2D). Along with increasing size, dark color in oil drops was gradually getting into light. The plasmids began disintegrating and many vesicles were formed from membranes of plasmids when the oil drops got to a certain size. The oil drops were packed with membranes of vesicles merged with each other (Figure 2E). The packed oil drops scattered in cytoplasm and trended to cellular membranes. The volatile compositions were released into intercellular space and air at the next step when the membranes of oil drops and cellular membranes merged (Figure 2F).
Discussion
There were different ideas about the accumulation mechanism of volatile compounds in cells. Most studies showed the oil cells were distributed in oil yielding plants. In previous research they believed that oil drops were produced in cytoplasm, then moved into oil vacuoles through the cupules after it was absorbed into the wall (Leemann, 1928). The oil drops in cytoplasm merged with oil vacuoles and was stored directly, or released out of cells and accumulated in cupule (Lehmann,1925; Maron et al, 1979). The other results show that oil was synthesized in plasmids, then moved into cytoplasm before getting into oil vacuoles (Amelunxen et al., 1969). The two paths of production and transportation of volatile oils were both found in oil cells of Magnolia denudate (Cai et al, 2001). In our study, the oil drops were observed in both cytoplasm and plasmids, but the two kinds were different. The oil drops in cytoplasm were packed with membrane, but the ones in intact plasmids were naked until plasmids disintegrated. Therefore, we believe that volatile oils synthesized in plasmids and were packed with membrane of disintegrated plasmids. Our research shows different results from other studies in the path of releasing volatile oil out of cells. Cupules and oil vacuoles were not found around oil cells, and membranes of the oil drops directly merged with cellular membranes before volatile oils released out of cell. The detailed mechanism for this remains to be further explained in future.
References
Amelunxen F., Gronau G. 1969. Elektronen-mikroskopische Untersuchungen an den Olzwllen von Acorus calamus L. Zeilschri/l fur Pflanzenphysiol, 60: 156-168.
Cai X., He Y., Hu Z. 2001. Ultrastructural studies on the oil cell development and oil accumulation in the Magnolia denudate. Journal of Northwest University (Natural Science Edition), 31(4): 351-354. (in Chinese)
Chang B., Hu Y., Xu Y., Zhang Q., Zhang W. 2007. Ultrastructures of petal surface of Osmanthus fragrans cultivars. Journal of Zhejiang Forestry College, 24(5): 533-537. (in Chinese)
Fan Z., Li J., Tian M., Li X., Ni S. 2006. Preliminary Studies of Aromatic Constituents between Three Species (Variation) of Camellia. Acta Horticulturae Sinica, 33(3): 592-596. (in Chinese)
Gu Y., Qian H. 2008. Analysis and mcroencapsulation of the essential oil from the flowers of Camellia sinensis. Food Research and Development, 29(1): 187-190. (in Chinese)
Leemann A. 1928. Das problem der Sekretzellen. Planta, 6: 216-233.
Lehmann C. 1925. Studien Uberden Bauunddie Entwicklungsgeschichte von Olzellen. Planta , 1: 343-373.
Maron R., Fahn A. 1979. Ultrastructure and development o f oil cells in Laurus nobilis L. leaves . Bot J Linn So c, 78: 31-40.
O’Brien Susan P., Loveys B. R., Grant W. J. R. 1996. Ultrastructure and function of floral nectarines of Chamelaucium uncinatum (Myrcaceae). Annals of Botany, 78: 189-196.
Smith M. T., Saks Y., Van Staden J. 1992. Ultrastructural changes in the petals of senescing flowers of Dianthus caryophyllus L.. Annals of Botany, 69: 277-285.
Song Xiao-hong, Peng L., Shi X., Dai S., Ye C. 2009. The aroma components from the flowers of Camellia renshanxiangiae by HS-SPME. Guihaia, 29(4): 561-563. (in Chinese)
Weston Pyke K A. 1999. Developmental ultrastructure of cells and platids in the petals of wallflower (Erysimum cheiri). Annals of Botany, 84: 763-769.
Web design by Tribal Systems
